Select a Size
| Pack Size | SKU | Availability | Price |
|---|
About This Item
grade
purum
Quality Level
assay
≥98.0% (GC)
form
crystals
bp
244-245 °C (lit.)
mp
53-56 °C, 54-56 °C (lit.)
density
1.324 g/mL at 25 °C (lit.)
functional group
chloro, ketone, phenyl
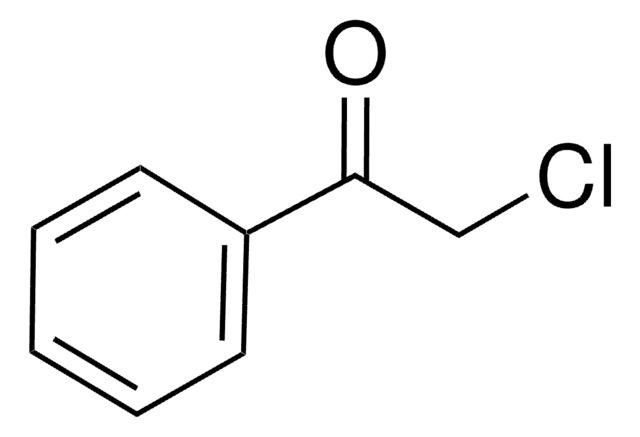
SMILES string
ClCC(=O)c1ccccc1
InChI
1S/C8H7ClO/c9-6-8(10)7-4-2-1-3-5-7/h1-5H,6H2
InChI key
IMACFCSSMIZSPP-UHFFFAOYSA-N
Gene Information
human ... PTPN6(5777)
General description
Application
1 of 1
This Item | |||
|---|---|---|---|
| assay ≥98.0% (GC) | assay 98% | assay 98% | assay ≥99.0% (GC) |
| Quality Level 200 | Quality Level 100 | Quality Level 100 | Quality Level 200 |
| density 1.324 g/mL at 25 °C (lit.) | density - | density - | density 0.851 g/mL at 25 °C (lit.) |
| mp 53-56 °C, 54-56 °C (lit.) | mp 48-51 °C (lit.) | mp 93-96 °C (lit.) | mp −25 °C (lit.) |
| bp 244-245 °C (lit.) | bp 135 °C/18 mmHg (lit.) | bp - | bp 51-52 °C (lit.) |
| form crystals | form solid | form solid | form - |
Still not finding the right product?
Explore all of our products under 2-Chloroacetophenone
signalword
Danger
Hazard Classifications
Acute Tox. 2 Oral - Acute Tox. 3 Dermal - Acute Tox. 3 Inhalation - Eye Dam. 1 - Resp. Sens. 1 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Faceshields, Gloves, type P2 (EN 143) respirator cartridges
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 77460-100G | 04061836686741 |
| 77460-500G | 04061836686758 |