94747
Vancomycin hydrochloride from Streptomyces orientalis
≥85% (Vancomycin B)
Synonym(s):
Vancomycin HCL
About This Item
Recommended Products
biological source
Streptomyces orientalis
Quality Level
Assay
≥85% (Vancomycin B)
form
powder
color
, off-white to brown or White to orange-brown
antibiotic activity spectrum
Gram-positive bacteria
Mode of action
cell wall synthesis | interferes
storage temp.
2-8°C
SMILES string
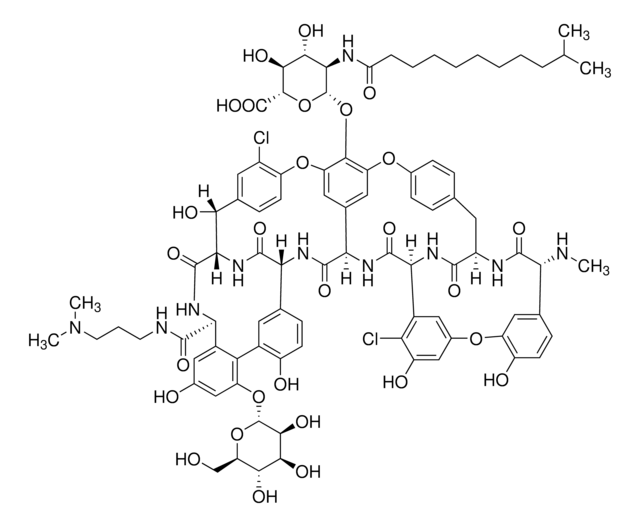
Cl[H].CO[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@@H]1O[C@H]2C[C@](C)(N)[C@@H](O)[C@@H](C)O2.CN[C@H](CC(C)C)C(=O)NC3[C@H](O)c4ccc(Oc5cc6Oc7ccc(cc7Cl)[C@@H](O)[C@H]8NC(=O)[C@H](NC(=O)[C@H](NC(=O)[C@H](CC(N)=O)NC3=O)c(c5)c6)c9ccc(O)c(c9)-c%10c(O)cc(O)cc%10[C@@H](NC8=O)C(O)=O)c(Cl)c4
InChI
1S/C66H75Cl2N9O24.ClH/c1-23(2)12-34(71-5)58(88)76-49-51(83)26-7-10-38(32(67)14-26)97-40-16-28-17-41(55(40)101-65-56(54(86)53(85)42(22-78)99-65)100-44-21-66(4,70)57(87)24(3)96-44)98-39-11-8-27(15-33(39)68)52(84)50-63(93)75-48(64(94)95)31-18-29(79)19-37(81)45(31)30-13-25(6-9-36(30)80)46(60(90)77-50)74-61(91)47(28)73-59(89)35(20-43(69)82)72-62(49)92;/h6-11,13-19,23-24,34-35,42,44,46-54,56-57,65,71,78-81,83-87H,12,20-22,70H2,1-5H3,(H2,69,82)(H,72,92)(H,73,89)(H,74,91)(H,75,93)(H,76,88)(H,77,90)(H,94,95);1H/t24-,34+,35-,42+,44-,46+,47+,48-,49+,50-,51+,52+,53+,54-,56+,57+,65-,66-;/m0./s1
InChI key
LCTORFDMHNKUSG-XTTLPDOESA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
Biochem/physiol Actions
Antimicrobial Spectrum: Active against Gram-positive bacteria
Packaging
Other Notes
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Resp. Sens. 1 - Skin Sens. 1
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
β-lactam antibacterials inhibit transpeptidase enzymes, preventing peptidoglycan assembly in both Gram-positive and Gram-negative bacteria.
β-lactam antibacterials inhibit transpeptidase enzymes, preventing peptidoglycan assembly in both Gram-positive and Gram-negative bacteria.
β-lactam antibacterials inhibit transpeptidase enzymes, preventing peptidoglycan assembly in both Gram-positive and Gram-negative bacteria.
β-lactam antibacterials inhibit transpeptidase enzymes, preventing peptidoglycan assembly in both Gram-positive and Gram-negative bacteria.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service