All Photos(2)
About This Item
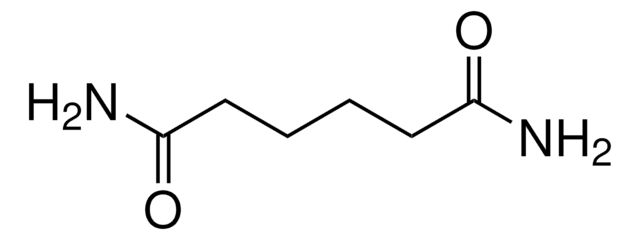
Linear Formula:
HO2C(CH2)5CO2H
CAS Number:
Molecular Weight:
160.17
Beilstein:
1210024
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
form
crystals
bp
212 °C/10 mmHg (lit.)
mp
103-105 °C (lit.)
SMILES string
OC(=O)CCCCCC(O)=O
InChI
1S/C7H12O4/c8-6(9)4-2-1-3-5-7(10)11/h1-5H2,(H,8,9)(H,10,11)
InChI key
WLJVNTCWHIRURA-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Pimelic acid, also known as heptanedioic acid, is a dicarboxylic acid commonly used as a precursor in the biosynthesis of biotin. It serves as a building block in the synthesis of various compounds , and it is used as a key precursor in the production of polyamides, polyesters, and other polymers.
Application
- Pimelic acid in microbial soil treatments: A 2023 study examined the use of pimelic acid in soil treatment, observing its impact on the microbial community structure and metabolites in bayberry plantations. This application illustrates its potential in agricultural biotechnology and soil remediation (Ren et al., 2023).
- Pharmaceutical applications of pimelic acid: A 2023 study involved pimelic acid in the preparation and physiochemical analysis of novel ciprofloxacin/dicarboxylic acid salts, showcasing its utility as a pharmaceutical intermediate (Hibbard et al., 2023).
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Kilogram Synthesis of Crebinostat
Yongjun et al.
Synthesis and Chemistry of Agrochemicals V, 48 (2016)
Bio-based, amorphous polyamides with tunable thermal properties
Van Velthoven et al.
European Polymer Journal, 66, 57-66 (2015)
Pimelic acid, the first precursor of the B acillus subtilis biotin synthesis pathway, exists as the free acid and is assembled by fatty acid synthesis
Manandhar et al.
Molecular Microbiology, 104, 595-607 (2017)
S Passi et al.
Acta dermato-venereologica. Supplementum, 143, 8-13 (1989-01-01)
Medium chain length dicarboxylic acids (DA) from C8 to C13 are competitive inhibitors of tyrosinase in vitro. The introduction of electron acceptor groups or electron donor groups into the 2 and/or the 8 position of the molecule enhances or reduces
T Niwa et al.
Clinica chimica acta; international journal of clinical chemistry, 99(1), 71-83 (1979-11-15)
(1) 2,4-Dimethyladipic acid was first identified in normal human urine using gas chromatography-mass spectrometry. Urinary excretion of 2,4-dimethyladipic acid in 7 healthy adults ranged from 4.9 mumol to 14 mumol per 24 h. (2) Succinic acid, adipic acid, 3-methyladipic acid
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service