292834
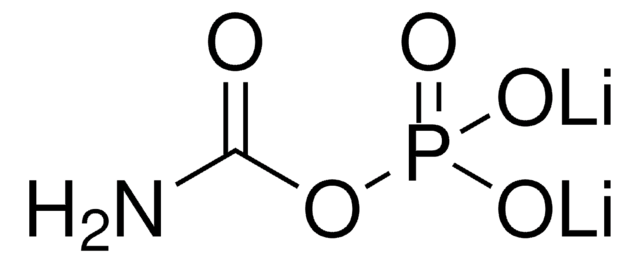
Ammonium carbamate
99%
Synonym(s):
Carbamic acid ammonium salt
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
H2NCO2NH4
CAS Number:
Molecular Weight:
78.07
Beilstein:
3914091
EC Number:
MDL number:
UNSPSC Code:
12352302
eCl@ss:
39033203
PubChem Substance ID:
NACRES:
NA.23
Recommended Products
vapor pressure
100 mmHg ( 26.7 °C)
Quality Level
Assay
99%
form
powder or chunks
expl. lim.
2 %
storage temp.
2-8°C
SMILES string
[H]N([H])[H].NC(O)=O
InChI
1S/CH3NO2.H3N/c2-1(3)4;/h2H2,(H,3,4);1H3
InChI key
BVCZEBOGSOYJJT-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
- Cooperative Carbon Dioxide Capture in Diamine-Appended Magnesium-Olsalazine Frameworks.: This article discusses a novel method for carbon dioxide capture using diamine-appended frameworks, where ammonium carbamate serves as a key chemical intermediate. This research underscores the environmental benefits of using ammonium carbamate in carbon capture technologies, contributing to efforts to mitigate climate change (Zhu et al., 2023).
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Dam. 1 - Skin Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Jo Sing Julia Tang et al.
Molecules (Basel, Switzerland), 25(21) (2020-11-08)
Glycans carry a vast range of functions in nature. Utilizing their properties and functions in form of polymers, coatings or glycan derivatives for various applications makes the synthesis of modified glycans crucial. Since amines are easy to modify for subsequent
Takayuki Furukawa et al.
Chembiochem : a European journal of chemical biology, 18(19), 1903-1909 (2017-08-06)
Although widely occurring lipid oxidation, which is triggered by reactive oxygen species (ROS), produces a variety of oxidized lipids, practical methods to efficiently analyze oxidized lipids remain elusive. Herein, it is shown that the glycoblotting platform can be used to
Akihiro Ohkubo et al.
Organic & biomolecular chemistry, 7(4), 687-694 (2009-02-06)
It was found that N-arylcarbamoyl and N-(phenylsulfonyl)carbamoyl (psc) groups could be effectively introduced onto the amino groups of deoxycytidine and deoxyadenosine derivatives and could be removed thermolytically. We succeeded in synthesizing DNA probes incorporating these thermo-removable protecting groups and developed
Robert Liu et al.
Waste management (New York, N.Y.), 29(11), 2842-2845 (2009-08-18)
The traditional burning process is used to recover copper from scrapped PC board (printed circuit board) but it causes serious environmental problems. In this research a new process was developed which not only prevents pollution problems, but also maximizes the
Yoshiaki Miura et al.
Analytical chemistry, 82(24), 10021-10029 (2010-11-17)
Glycoblotting, high throughput method for N-glycan enrichment analysis based on the specific chemical ligation between aminooxy/hydrazide-polymers/solids and reducing N-glycans released from whole serum and cellular glycoproteins, was proved to be feasible for selective enrichment analysis of O-glycans of common (mucin)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service






![[Bis(trifluoroacetoxy)iodo]benzene 97%](/deepweb/assets/sigmaaldrich/product/structures/238/293/71fcde9a-4afb-4cf5-9c22-8d8d68bf1ba4/640/71fcde9a-4afb-4cf5-9c22-8d8d68bf1ba4.png)