250287
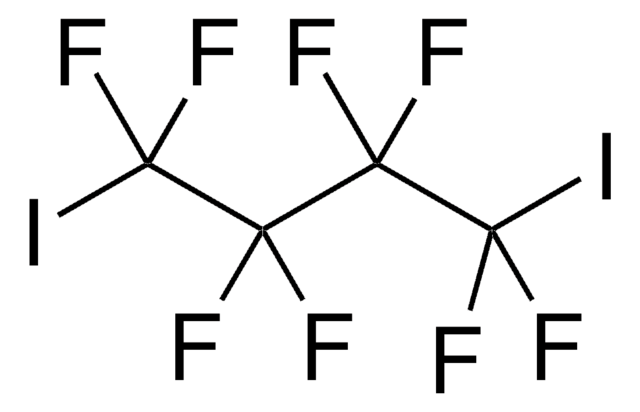
1,10-Diiododecane
95%
Synonym(s):
Decamethylene diiodide
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
I(CH2)10I
CAS Number:
Molecular Weight:
394.07
Beilstein:
1738614
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
95%
form
liquid
bp
197-200 °C/12 mmHg (lit.)
mp
33-35 °C (lit.)
functional group
iodo
SMILES string
ICCCCCCCCCCI
InChI
1S/C10H20I2/c11-9-7-5-3-1-2-4-6-8-10-12/h1-10H2
InChI key
CKJCTZAIDVFHCX-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Electrochemical reduction of 1,10-diiododecane at Hg cathode in dimethylformamide containing tetramethylammonium perchlorate has been investigated.
Application
1,10-Diiododecane has been used as:
- an alkylating agent in the synthesis of symmetrical and unsymmetrical bis-cryptophanes
- crosslinking reagent for tetrabutylammonium polygalacturonic acid
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
235.4 °F - closed cup
Flash Point(C)
113 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Gelation of chemically cross-linked polygalacturonic acid derivatives.
Matricardi P, et al.
Carbohydrate Polymers, 27(3), 215-220 (1995)
Electrochemical reduction of 1, 10-dihalodecanes at mercury cathodes in dimethylformamide.
Bart JC and Peters DG.
J. Electroanal. Chem. Interfac. Electrochem., 280(1), 129-144 (1990)
Magali Darzac et al.
Chemical communications (Cambridge, England), (1)(1), 48-49 (2002-07-18)
After deprotection with a palladium catalyst, mono-allylated cryptophane-A (1, 2) and cryptophane-E (3) gave the new cryptophanols 4, 5 and 6, respectively, which are important key compounds for the preparation of monofunctionalized cryptophanes as well as for the design of
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service