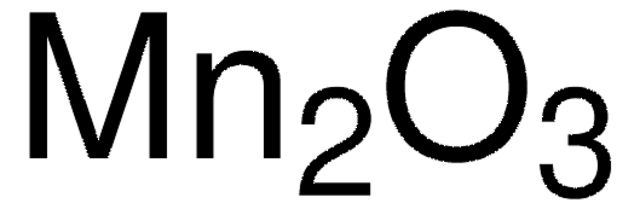215880
Manganese(III) acetate dihydrate
97%
Synonym(s):
Manganese triacetate dihydrate
About This Item
Recommended Products
Quality Level
Assay
97%
form
powder or chunks
reaction suitability
core: manganese
SMILES string
O.O.CC(=O)O[Mn](OC(C)=O)OC(C)=O
InChI
1S/3C2H4O2.Mn.2H2O/c3*1-2(3)4;;;/h3*1H3,(H,3,4);;2*1H2/q;;;+3;;/p-3
InChI key
ONJSLAKTVIZUQS-UHFFFAOYSA-K
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
- A precursor in the synthesis of manganese oxide (Mn3O4) nanostructures, which are employed as anode materials in lithium-ion batteries.
- A manganese source in the sol-gel synthesis of Mn-doped ZnO thin films. The incorporation of manganese ions into the ZnO lattice is essential for modifying the electronic and optical properties of the films.
- A precursor for the synthesis of manganese oxide nanoparticles using a sol-gel process. These nanoparticles are evaluated for their performance in supercapacitor applications.
- A mild and selective oxidizing agent. Catalyzes allylic oxidation of a variety of alkenes in the presence of tert-butylhydroperoxide. Reagent used for radical cyclizations and α-keto-acetoxylation.
Physical form
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Personal Protective Equipment
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Oxidation and reduction reactions are some of the most common transformations encountered in organic synthesis, and are some of the organic chemist’s most powerful tools for creating novel products. Below is a list of the most commonly used oxidizing and reducing agents currently available in our catalog.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service