T7383
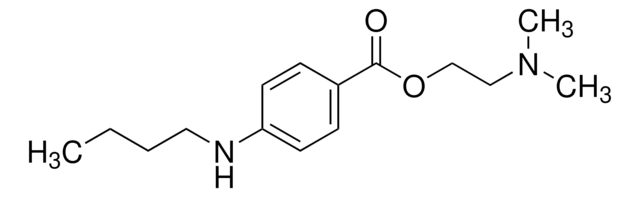
Tetracaine
≥98% (TLC)
Sinónimos:
4-(Butylamino)benzoic acid 2-(dimethylamino)ethyl ester
About This Item
Productos recomendados
origen biológico
synthetic (organic)
Nivel de calidad
Ensayo
≥98% (TLC)
Formulario
powder
técnicas
toxicology assay: suitable
temp. de almacenamiento
2-8°C
cadena SMILES
CCCCNc1ccc(cc1)C(=O)OCCN(C)C
InChI
1S/C15H24N2O2/c1-4-5-10-16-14-8-6-13(7-9-14)15(18)19-12-11-17(2)3/h6-9,16H,4-5,10-12H2,1-3H3
Clave InChI
GKCBAIGFKIBETG-UHFFFAOYSA-N
Información sobre el gen
human ... SCN10A(6336) , SCN11A(11280) , SCN1A(6323) , SCN2A(6326) , SCN3A(6328) , SCN4A(6329) , SCN5A(6331) , SCN7A(6332) , SCN8A(6334) , SCN9A(6335)
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
Descripción general
Aplicación
Acciones bioquímicas o fisiológicas
Nota de preparación
Palabra de señalización
Danger
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Acute Tox. 3 Oral - Carc. 2 - Skin Sens. 1
Código de clase de almacenamiento
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
Clase de riesgo para el agua (WGK)
WGK 3
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico