643777
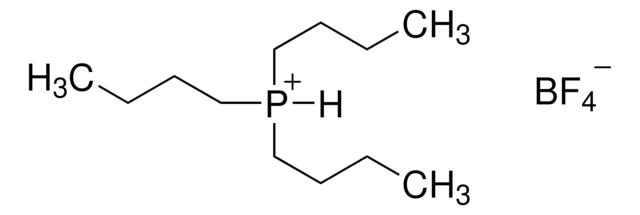
Di-tert-butyl(methyl)phosphonium tetrafluoroborate
97%
Sinónimos:
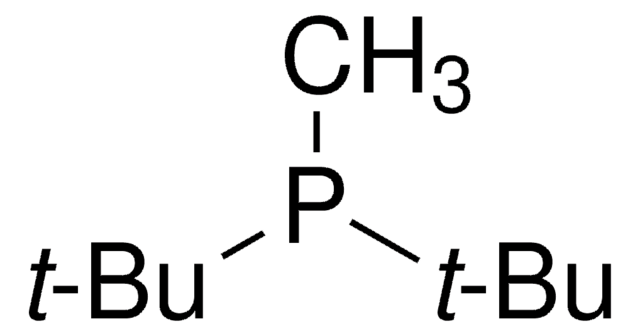
(t-Bu)2PMeHBF4, Bis(1,1-dimethylethyl)methylphosphine tetrafluoroborate
About This Item
Productos recomendados
Quality Level
assay
97%
form
solid
reaction suitability
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reaction type: Heck Reaction
reaction type: Hiyama Coupling
reaction type: Negishi Coupling
reaction type: Sonogashira Coupling
reaction type: Stille Coupling
reaction type: Suzuki-Miyaura Coupling
reagent type: ligand
reaction type: Cross Couplings
pH
1.79 (1% in solution)
mp
>230 °C (lit.)
functional group
phosphine
SMILES string
F[B-](F)(F)F.C[PH+](C(C)(C)C)C(C)(C)C
InChI
1S/C9H21P.BF4/c1-8(2,3)10(7)9(4,5)6;2-1(3,4)5/h1-7H3;/q;-1/p+1
InChI key
BRDLRXCAHKUWJS-UHFFFAOYSA-O
¿Está buscando productos similares? Visita Guía de comparación de productos
Application
signalword
Danger
hcodes
Hazard Classifications
Eye Dam. 1 - Skin Corr. 1B
Storage Class
8A - Combustible corrosive hazardous materials
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico


![[Pd(OAc)2]3 reagent grade, 98%](/deepweb/assets/sigmaaldrich/product/structures/508/249/99a0ef2c-b77c-4d73-8ed9-0cca05b6b41f/640/99a0ef2c-b77c-4d73-8ed9-0cca05b6b41f.png)




