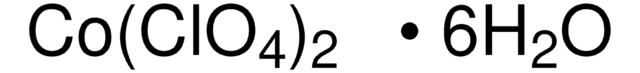401439
Zinc perchlorate hexahydrate
Sinónimos:
Perchloric acid zinc hydrate
About This Item
Productos recomendados
form
crystals and lumps
Quality Level
reaction suitability
reagent type: oxidant
SMILES string
O.O.O.O.O.O.[Zn++].[O-]Cl(=O)(=O)=O.[O-]Cl(=O)(=O)=O
InChI
1S/2ClHO4.6H2O.Zn/c2*2-1(3,4)5;;;;;;;/h2*(H,2,3,4,5);6*1H2;/q;;;;;;;;+2/p-2
Inchi Key
PADPILQDYPIHQQ-UHFFFAOYSA-L
General description
Application
- For acylation of electron-deficient phenols, sterically hindered alcohols and amines.
- For the synthesis of 2-hydroxy sulfides by the opening of epoxide rings with thiols.
- For solvent‐free condensation of carboxylic acids with alcohols.
- An electrolyte for aqueous Zinc batteries.
- A precursor for the synthesis of highly fluorescent ZnSe quantumdots.
signalword
Danger
hcodes
Hazard Classifications
Ox. Sol. 2 - Skin Corr. 1B
Storage Class
5.1A - Strongly oxidizing hazardous materials
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Artículos
Colloidal quantum dots (CQDs) are semiconducting crystals of only a few nanometers (ca. 2–12 nm) coated with ligand/surfactant molecules to help prevent agglomeration.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico


![Zinc di[bis(trifluoromethylsulfonyl)imide] 95%](/deepweb/assets/sigmaaldrich/product/structures/336/073/952daadd-0a7c-4bec-bbaf-442a24c62161/640/952daadd-0a7c-4bec-bbaf-442a24c62161.png)