304484
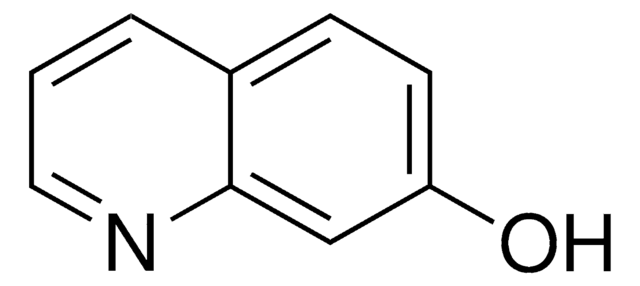
6-Hydroxyquinoline
95%
Sinónimos:
6-Quinolinol, 6-Hydroxyquinoline
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula empírica (notación de Hill):
C9H7NO
Número de CAS:
Peso molecular:
145.16
Beilstein/REAXYS Number:
113196
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Productos recomendados
Quality Level
assay
95%
mp
188-190 °C (lit.)
SMILES string
Oc1ccc2ncccc2c1
InChI
1S/C9H7NO/c11-8-3-4-9-7(6-8)2-1-5-10-9/h1-6,11H
InChI key
OVYWMEWYEJLIER-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
General description
6-Hydroxyquinoline is an ideal photoacid system for exploring excited-state proton transfer (ESPT) reactions. The excited-state proton transfer and geminate recombination of 6-hydroxyquinoline encaged in catalytic Na+-exchanged faujasite zeolites X and Y have been explored by measuring steady-state and picosecond time-resolved spectra.
Application
6-Hydroxyquinoline was used in synthesis of 2,6-substituted-benzo[d]thiazole analogs and 2,4-substituted-benzo[d]thiazole analogs.
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Katharigatta Narayanaswamy Venugopala et al.
European journal of medicinal chemistry, 65, 295-303 (2013-06-04)
A novel and efficient one pot synthesis was developed for 2,6-substituted-benzo[d]thiazole analogues 4a-k and 2,4-substituted-benzo[d]thiazole analogues 4l-pvia three component condensation reaction of substituted arylaldehyde, 2-amino-6-halo/4-methyl-benzo[d]thiazole and 2-naphthol or 6-hydroxyquinoline in presence of 10% w/v NaCl in water by microwave method.
Yu-Hui Liu et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 128, 280-284 (2014-04-01)
6-Hydroxyquinoline (6HQ) is an ideal photoacid system for exploring excited-state proton transfer (ESPT) reactions. We have previously (Mahata et al. (2002)) shown that the ESPT reaction between 6HQ and trimethylamine (TMA) leads to an "unusual" emission in the 440-450 nm
G Bott et al.
Biological chemistry Hoppe-Seyler, 372(6), 381-383 (1991-06-01)
Two strains, using 6-hydroxyquinoline as sole source of energy, carbon and nitrogen, have been isolated. These bacteria, designated 31/1 Fa1 and 31/2 A1, are also able to degrade quinoline. According to their physiological properties strain 31/1 Fa1 has been identified
Selective phenolic acylation of 10-hydroxycamptothecin using poly (ethylene glycol) carboxylic acid.
Richard B Greenwald et al.
Bioorganic & medicinal chemistry letters, 13(3), 577-580 (2003-02-05)
Selective acylation of the phenolic hydroxyl group of 10-hydroxycamptothecin has been accomplished using phenyl dichlorophosphate. Additional modification of the 10-OH as an ether permits a 20-acyl derivative to be synthesized. This result along with data from a 6-hydroxyquinoline model strongly
Anna Michta et al.
Acta crystallographica. Section C, Crystal structure communications, 65(Pt 2), o66-o69 (2009-02-05)
The title compound, C(9)H(7)NO, has two symmetry-independent molecules in the asymmetric unit, which have different conformations of the hydroxy group with respect to the quinoline ring. One of the molecules adopts a cis conformation, while the other shows a trans
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico