160660
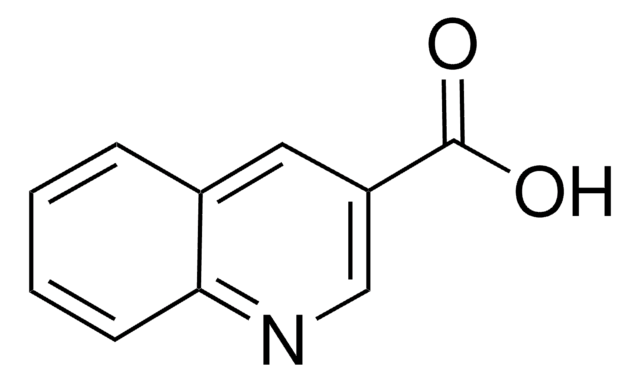
Quinaldic acid
98%
Sinónimos:
2-Quinolinecarboxylic acid
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula empírica (notación de Hill):
C10H7NO2
Número de CAS:
Peso molecular:
173.17
Beilstein:
126322
Número CE:
Número MDL:
Código UNSPSC:
12352100
ID de la sustancia en PubChem:
NACRES:
NA.22
Productos recomendados
Nivel de calidad
Ensayo
98%
Formulario
solid
mp
156-158 °C (lit.)
grupo funcional
carboxylic acid
cadena SMILES
OC(=O)c1ccc2ccccc2n1
InChI
1S/C10H7NO2/c12-10(13)9-6-5-7-3-1-2-4-8(7)11-9/h1-6H,(H,12,13)
Clave InChI
LOAUVZALPPNFOQ-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Descripción general
Quinaldic acid is also referred as quinoline-2-carboxylic acid. Microwave-assisted preparation of substituted anilides of quinaldic acid has been reported. It inhibits the oxidation of pyruvate, α-ketoglutarate, glutamate and citrate in rat liver mitochondria. Quinaldic acid is a metabolite of tryptophan degradation and inhibits the gluconeogenesis in perfused livers.
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Felipe Lombó et al.
Chembiochem : a European journal of chemical biology, 7(2), 366-376 (2006-01-13)
Thiocoraline is a thiodepsipeptide antitumor compound produced by two actinomycetes Micromonospora sp. ACM2-092 and Micromonospora sp. ML1, isolated from two marine invertebrates (a soft coral and a mollusc) found of the Indian Ocean coast of Mozambique. By using oligoprimers derived
B R Bochner et al.
Journal of bacteriology, 143(2), 926-933 (1980-08-01)
A simple technique has been devised that allows direct plate selection of tetracycline-sensitive clones from a predominantly tetracycline-resistant population. The technique is especially useful in genetic methodologies based on the use of tetracycline resistance transposons, such as Tn10. Potential uses
Alleyn T Plowright et al.
Chemistry & biology, 9(5), 607-618 (2002-05-29)
Saframycin A (SafA) is a natural product that inhibits human cancer cell proliferation. Its synthetic analog, QAD, is a more potent inhibitor of these cells. SafA does not affect wild-type yeast, but it does inhibit growth of the strain CCY333
Stereocontrolled synthesis of the quinaldic acid macrocyclic system of thiostrepton.
K C Nicolaou et al.
Angewandte Chemie (International ed. in English), 41(11), 1937-1940 (2002-06-03)
K Jhamandas et al.
Brain research, 529(1-2), 185-191 (1990-10-08)
Certain products of tryptophan metabolism interact with excitatory amino acid receptors to produce or protect against excitotoxicity. In this study, the action of several tryptophan metabolites, yielded by the kynurenine pathway, on cortical cholinergic toxicity was evaluated following focal injection
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico