159425
9-Chloroacridine
97%
Sinónimos:
9-Chloroacridine
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
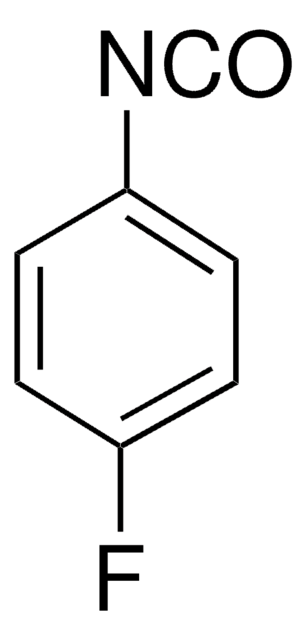
Fórmula empírica (notación de Hill):
C13H8ClN
Número de CAS:
Peso molecular:
213.66
Número CE:
Número MDL:
Código UNSPSC:
12352100
ID de la sustancia en PubChem:
NACRES:
NA.22
Productos recomendados
Nivel de calidad
Ensayo
97%
Formulario
solid
mp
116-120 °C (lit.)
grupo funcional
chloro
cadena SMILES
Clc1c2ccccc2nc3ccccc13
InChI
1S/C13H8ClN/c14-13-9-5-1-3-7-11(9)15-12-8-4-2-6-10(12)13/h1-8H
Clave InChI
BPXINCHFOLVVSG-UHFFFAOYSA-N
Aplicación
9-Chloroacridine was employed as chromogenic reagent in the spectrophotometric method for the quantitative determination of dapsone. It was also used in the synthesis of:
- series of novel chalcones bearing acridine moiety attached to the amino group in their ring A
- new acridine derivatives
- 9-phenoxyacridine and 4-phenoxyfuro[2,3-b]quinoline derivatives
Palabra de señalización
Warning
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Órganos de actuación
Respiratory system
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Paula Bosch et al.
Materials (Basel, Switzerland), 12(18) (2019-09-22)
A second-generation poly(propylene imine) dendrimer modified with acridine and its Cu(II) complex have been synthesized for the first time. It has been found that two copper ions form complexes with the nitrogen atoms of the dendrimeric core by coordinate bonds.
Jelena Rupar et al.
Bioelectrochemistry (Amsterdam, Netherlands), 135, 107579-107579 (2020-06-14)
The electrochemical behavior of 9-chloroacridine (9Cl-A), a precursor molecule for synthesis of acridine derivatives with cytostatic activity, is a complex, pH-dependent, diffusion-controlled irreversible process. Oxidation of 9Cl-A initiates with the formation of a cation radical monomer, continues via the formation
V Tomar et al.
European journal of medicinal chemistry, 45(2), 745-751 (2009-12-22)
A series of novel chalcones bearing acridine moiety attached to the amino group in their ring A have been synthesized through noncatalyzed nucleophilic aromatic substitution reaction between various 3'-aminochalcone or 4'-aminochalcones and 9-chloroacridine. The synthesized chalcone derivatives have been characterized
Arumugasamy Elangovan et al.
Organic & biomolecular chemistry, 2(21), 3113-3118 (2004-10-27)
Electrogenerated chemiluminescence (ECL) of six new ethyne-based acridine derivatives (1-6) has been studied. The new acridine derivatives were synthesized by cross-coupling of 9-chloroacridine and corresponding donor-substituted phenylethynes under modified Sonogashira conditions. The donor groups were varied in the order of
I Shoukrallah et al.
Die Pharmazie, 45(9), 675-677 (1990-09-01)
A spectrophotometric method for the quantitative determination of dapsone (1) has been developed through a condensation reaction of 9-chloroacridine as a chromogen and the amino groups of 1. The reaction variables were investigated and optimized. The resultant colored products is
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico