PHR1238
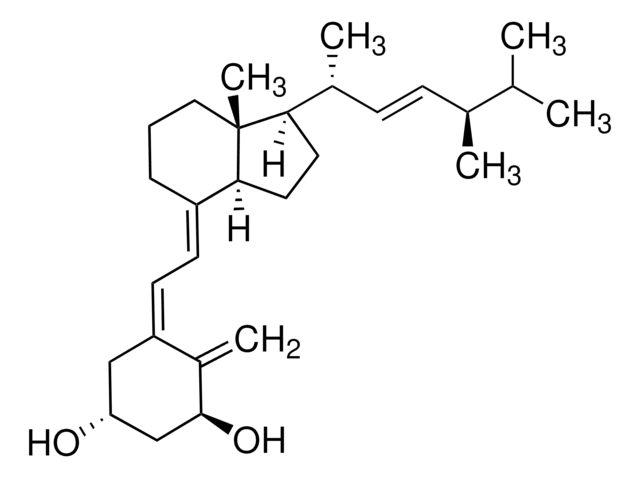
Ergocalciferol
Pharmaceutical Secondary Standard; Certified Reference Material
Sinónimos:
Ergocalciferol, Calciferol, Ercalciol, Ergosterol irradiated, Irradiated ergosterol, Vitamin D2
About This Item
Productos recomendados
grado
certified reference material
pharmaceutical secondary standard
Nivel de calidad
Agency
traceable to BP 788
traceable to Ph. Eur. E0900000
traceable to USP 1239005
familia API
ergocalciferol
CofA
current certificate can be downloaded
técnicas
HPLC: suitable
gas chromatography (GC): suitable
mp
114-118 °C (lit.)
aplicaciones
clinical testing
Formato
neat
temp. de almacenamiento
-10 to -25°C
cadena SMILES
CC(C)[C@@H](C)/C=C/[C@@H](C)[C@@]1([H])CC[C@@]([C@]1(C)CCC/2)([H])C2=C\C=C(C[C@@H](O)CC3)/C3=C
InChI
1S/C28H44O/c1-19(2)20(3)9-10-22(5)26-15-16-27-23(8-7-17-28(26,27)6)12-13-24-18-25(29)14-11-21(24)4/h9-10,12-13,19-20,22,25-27,29H,4,7-8,11,14-18H2,1-3,5-6H3/b10-9+,23-12+,24-13-/t20-,22+,25-,26+,27-,28+/m0/s1
Clave InChI
MECHNRXZTMCUDQ-RKHKHRCZSA-N
Información sobre el gen
human ... VDR(7421)
¿Está buscando productos similares? Visita Guía de comparación de productos
Descripción general
Aplicación
Nota de análisis
Otras notas
Nota al pie de página
Producto relacionado
Palabra de señalización
Danger
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Acute Tox. 2 Inhalation - Acute Tox. 3 Dermal - Acute Tox. 3 Oral - STOT RE 1 Oral
Código de clase de almacenamiento
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Elija entre una de las versiones más recientes:
Certificados de análisis (COA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Documentos section.
Si necesita más asistencia, póngase en contacto con Atención al cliente
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Protocolos
Ascentis Express C18 is suitable for routine analysis of fat-soluble vitamins D2 and D3, providing optimized chromatograms.
Ascentis Express C18 is suitable for routine analysis of fat-soluble vitamins D2 and D3, providing optimized chromatograms.
Ascentis Express C18 is suitable for routine analysis of fat-soluble vitamins D2 and D3, providing optimized chromatograms.
Separation of Ergocalciferol (D2), analytical standard; Cholecalciferol (D3), analytical standard
Active Filters
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico