656577
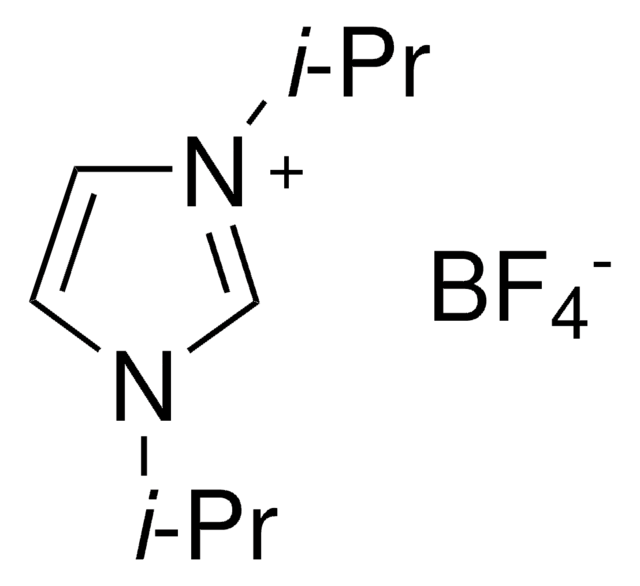
1,3-Diisopropylimidazolium chloride
97%
Sinónimos:
N,N′-(Isopropyl)imidazolium chloride
About This Item
Productos recomendados
Quality Level
assay
97%
form
solid
reaction suitability
reagent type: catalyst
greener alternative product characteristics
Catalysis
Learn more about the Principles of Green Chemistry.
sustainability
Greener Alternative Product
mp
182-186 °C
greener alternative category
SMILES string
[Cl-].CC(C)n1cc[n+](c1)C(C)C
InChI
1S/C9H17N2.ClH/c1-8(2)10-5-6-11(7-10)9(3)4;/h5-9H,1-4H3;1H/q+1;/p-1
InChI key
DOFXKPAOJLLPII-UHFFFAOYSA-M
General description
Application
- 1,3-Diisopropylimidazole-2-thione by reacting with sulfur in the presence of potassium carbonate.
- (C9H7)NiCl(1,3-diisopropylimidazol-2- ylidene) by reacting with bis-indenyl nickel-(II).
- Ruthenium N-heterocyclic carbene complexes, which are efficient catalysts for the amidation of primary alcohols and amines.
Amide Synthesis from Alcohols and Amines by the Extrusion of Dihydrogen
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Protocolos
Acetylene chemistry's versatile applications from organic synthesis to bioorganic chemistry demand efficient synthesis methods.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico