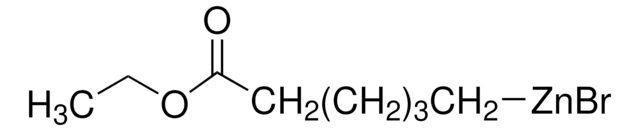513830
Indole-5-carboxaldehyde
98%
Sinónimos:
5-Formylindole
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula empírica (notación de Hill):
C9H7NO
Número de CAS:
Peso molecular:
145.16
Número MDL:
Código UNSPSC:
12352100
ID de la sustancia en PubChem:
NACRES:
NA.22
Productos recomendados
Ensayo
98%
mp
100-103 °C (lit.)
grupo funcional
aldehyde
cadena SMILES
O=Cc1ccc2[nH]ccc2c1
InChI
1S/C9H7NO/c11-6-7-1-2-9-8(5-7)3-4-10-9/h1-6,10H
Clave InChI
ADZUEEUKBYCSEY-UHFFFAOYSA-N
Categorías relacionadas
Aplicación
Indole-5-carboxaldehyde can be used as a reactant in the:
- Preparation of curcumin derivatives as anti-proliferative & anti-inflammatory agents
- Preparation of analogs of botulinum neurotoxin serotype A protease inhibitors
- Stereoselective synthesis of dibenzylideneacetone derivatives as β-amyloid imaging probes
- Synthesis of para-para stilbenophanes by McMurry coupling
- Stereoselective synthesis of heteroaromatic (E)-α,β-unsaturated ketones from aldehydes
- Structure-based drug design of aurora kinase A inhibitors
- Preparation of 5-indolyl linked 15- and 18-membered azacrown ethers to study their cation-π interactions.
- Preparation of bibenzimidazole derivatives substituted 5-indolyl moiety in the study of inhibition of topoisomerase I activity.
- Synthesis of (5-(4-(3,4,5-trimethoxybenzoyl)-1H-imidazol-2-yl)-1H-indol-2-yl)(3,4,5-trimethoxyphenyl)methanone and radioiodinated indolochalcone.
Palabra de señalización
Warning
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Órganos de actuación
Respiratory system
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Raquel Álvarez et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 17(12), 3406-3419 (2011-02-24)
The synthesis of a new family of methoxy-substituted [2.7]- and [2.8]paracyclophanes linked by 3-oxapentamethylene-1,5-dioxy and hexamethylene-1,6-dioxy bridges has been carried out by using the McMurry methodology. Related indole compounds were also synthesised. Olefin-to-diol ratios depended on the bridge length, the
Synthesis, QSAR and hypoglycemic activity of substituted 2,4-thiazolidinedione derivatives
Bahara, R. S.; Kulkarni, V. M.
Der Pharma Chemica, 3, 164-164 (2011)
Todd J Eckroat et al.
Beilstein journal of organic chemistry, 9, 1012-1044 (2013-06-15)
The number of people suffering from Alzheimer's disease (AD) is expected to increase dramatically in the coming years, placing a huge burden on society. Current treatments for AD leave much to be desired, and numerous research efforts around the globe
S Jin et al.
Bioorganic & medicinal chemistry letters, 10(8), 719-723 (2000-04-27)
A series of 2'-heterocyclic derivatives of 5-phenyl-2,5'-1H-bibenzimidazoles were evaluated for topoisomerase I poisoning activity and cytotoxicity. Topo I poisoning activity was associated with 2'-derivatives that possessed a hydrogen atom capable of hydrogen bond formation, suggesting that the interatomic distances between
Paul R Carlier et al.
The Journal of organic chemistry, 67(17), 6256-6259 (2002-08-17)
Tryptophan 1 (Trp) is superior to all other naturally occurring peptide residues in its ability to bind cations (the cation-pi interaction). In an effort to expand the toolbox of Trp-like amino acids, in this note we report catalytic asymmetric syntheses
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico