91862
Trimethyl(trifluoromethyl)silane solution
2 M in THF
Sinonimo/i:
(Trifluoromethyl)trimethylsilane, Ruppert′s reagent, Ruppert-Prakash reagent, TFMTMS, Trifluoromethyltrimethylsilane
About This Item
Prodotti consigliati
Forma fisica
liquid
Impiego in reazioni chimiche
reaction type: C-C Bond Formation
IVD
for in vitro diagnostic use
Concentrazione
2 M in THF
Indice di rifrazione
n20/D 1.386
Densità
0.91 g/mL at 20 °C
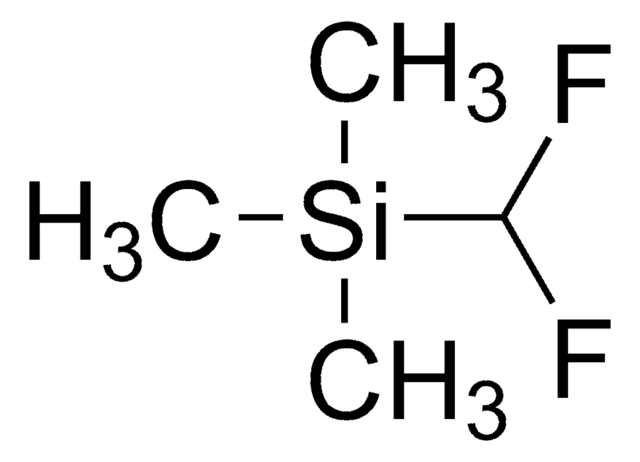
Stringa SMILE
C[Si](C)(C)C(F)(F)F
InChI
1S/C4H9F3Si/c1-8(2,3)4(5,6)7/h1-3H3
MWKJTNBSKNUMFN-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Descrizione generale
Applicazioni
- Silver-mediated C-H trifluoromethylation of arenes
- Preparation of trifluoromethyl ketone analog of L-arginine having contrasting inhibitory activity against human arginase I and histone deacetylase 8
- Organocatalyzed regio- and enantioselective allylic trifluoromethylation of Morita-Baylis-Hillman adducts
- Palladium-catalyzed oxidative trifluoromethylation of indoles
- Preparation of 5-HT1A antagonists
- Used as difluorocarbene source
- Conversion of aromatic aldehydes to difluoromethylated products.
- C-H trifluoromethylation of arenes, terminal alkynes, tertiary amines, heteroarenes, allylic, and terminal alkenes using metal catalyst or metal free oxidative trifluoromethylation reaction.
It can also be used in trifluoromethylation of:
- Non-activated aldimines.
- Heterocumulenes.
- Azomethine imines.
Avvertenze
Danger
Indicazioni di pericolo
Classi di pericolo
Acute Tox. 4 Oral - Carc. 2 - Eye Irrit. 2 - Flam. Liq. 2 - STOT SE 3
Organi bersaglio
Respiratory system
Rischi supp
Codice della classe di stoccaggio
3 - Flammable liquids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
1.4 °F
Punto d’infiammabilità (°C)
-17 °C
Dispositivi di protezione individuale
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Certificati d'analisi (COA)
Cerca il Certificati d'analisi (COA) digitando il numero di lotto/batch corrispondente. I numeri di lotto o di batch sono stampati sull'etichetta dei prodotti dopo la parola ‘Lotto’ o ‘Batch’.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.