1278007
USP
Fluorometholone
United States Pharmacopeia (USP) Reference Standard
Synonyme(s) :
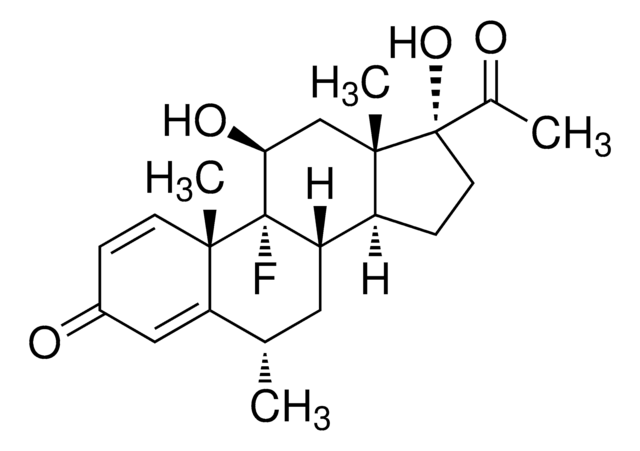
11β,17α-Dihydroxy-9-fluoro-6-methyl-1,4-pregnadiene-3,20-dione
About This Item
Produits recommandés
Qualité
pharmaceutical primary standard
Famille d'API
fluorometholone
Fabricant/nom de marque
USP
Application(s)
pharmaceutical (small molecule)
Format
neat
Chaîne SMILES
C[C@H]1C[C@H]2[C@@H]3CC[C@](O)(C(C)=O)[C@@]3(C)C[C@H](O)[C@]2(F)[C@@]4(C)C=CC(=O)C=C14
InChI
1S/C22H29FO4/c1-12-9-17-15-6-8-21(27,13(2)24)20(15,4)11-18(26)22(17,23)19(3)7-5-14(25)10-16(12)19/h5,7,10,12,15,17-18,26-27H,6,8-9,11H2,1-4H3/t12-,15-,17-,18-,19-,20-,21-,22-/m0/s1
Clé InChI
FAOZLTXFLGPHNG-KNAQIMQKSA-N
Informations sur le gène
human ... NR3C1(2908)
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Description générale
Application
- Fluorometholone Acetate
- Fluorometholone Ophthalmic Suspension
- Hydrocortisone Hemisuccinate
- Hydrocortisone Sodium Succinate for Injection
- Methylprednisolone Hemisuccinate
- Methylprednisolone Sodium Succinate for Injection
Actions biochimiques/physiologiques
Remarque sur l'analyse
Autres remarques
Mention d'avertissement
Warning
Mentions de danger
Conseils de prudence
Classification des risques
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique