792551
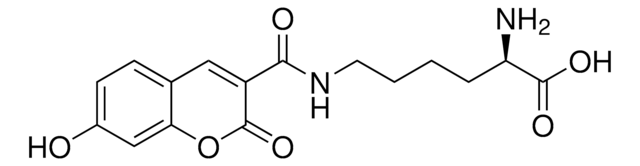
H-4-(7-Hydroxy-4-coumarinyl)-Abu-OH
98% (HPLC)
Synonyme(s) :
(S)-2-Amino-4-(7-hydroxy-2-oxo-2H-chromen-4-yl)butanoic acid, Schultz Fluorescent UAA
Se connecterpour consulter vos tarifs contractuels et ceux de votre entreprise/organisme
About This Item
Formule empirique (notation de Hill):
C13H13NO5
Poids moléculaire :
263.25
Code UNSPSC :
12352111
Nomenclature NACRES :
NA.22
Produits recommandés
Niveau de qualité
Pureté
98% (HPLC)
Forme
solid
Activité optique
[α]22/D +8.0°, c = 0.5% in 1 M NaOH
Pf
276-281 °C
Température de stockage
15-25°C
Application
The courmarin-based unnatural amino acid is an effective fluorophore; which has been incoporated into several studies of protein interactions.
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Ishu Saraogi et al.
Journal of the American Chemical Society, 133(38), 14936-14939 (2011-08-30)
As newly synthesized proteins emerge from the ribosome, they interact with a variety of cotranslational cellular machineries that facilitate their proper folding, maturation, and localization. These interactions are essential for proper function of the cell, and the ability to study
Douglas D Young et al.
Bioorganic & medicinal chemistry letters, 21(24), 7502-7504 (2011-11-02)
The site-specific incorporation of unnatural amino acids (UAAs) into proteins in bacteria is made possible by the evolution of aminoacyl-tRNA synthetases that selectively recognize and aminoacylate the amino acid of interest. Recently we have discovered that some of the previously
Jiangyun Wang et al.
Journal of the American Chemical Society, 128(27), 8738-8739 (2006-07-06)
The fluorescent amino acid l-(7-hydroxycoumarin-4-yl) ethylglycine 1 has been genetically encoded in E. coli in response to the amber TAG codon. Because of its high fluorescence quantum yield, relatively large Stoke's shift, and sensitivity to both pH and polarity, this
Timo Koopmans et al.
Bioorganic & medicinal chemistry, 21(2), 553-559 (2012-12-15)
Incorporation of the unnatural amino acid L-(7-hydroxycoumarin-4-yl)ethylglycine (7-HC) is a powerful and reliable approach for the preparation of fluorescently labeled proteins. The growing popularity of this valuable amino acid prompted us to pursue an improved protocol for its synthetic preparation.
Shengxi Chen et al.
Journal of the American Chemical Society, 135(35), 12924-12927 (2013-08-15)
Two fluorescent amino acids, including the novel fluorescent species 4-biphenyl-l-phenylalanine (1), have been incorporated at positions 17 and 115 of dihydrofolate reductase (DHFR) to enable a study of conformational changes associated with inhibitor binding. Unlike most studies involving fluorescently labeled
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique