90520
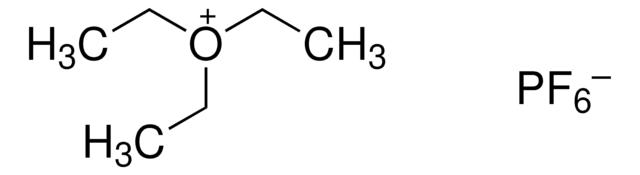
Triethyloxonium tetrafluoroborate
≥97.0% (T)
Synonym(s):
Et3OBF4, Meerwein′s reagent, Triethyloxonium fluoroborate
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
(C2H5)3O(BF4)
CAS Number:
Molecular Weight:
189.99
Beilstein:
3598090
MDL number:
UNSPSC Code:
12352001
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
≥97.0% (T)
form
crystals
contains
1-3% ether as stabilizer
solubility
methylene chloride: 20 mg/mL, clear, colorless
storage temp.
2-8°C
SMILES string
F[B-](F)(F)F.CC[O+](CC)CC
InChI
1S/C6H15O.BF4/c1-4-7(5-2)6-3;2-1(3,4)5/h4-6H2,1-3H3;/q+1;-1
InChI key
IYDQMLLDOVRSJJ-UHFFFAOYSA-N
Application
Triethyloxonium tetrafluoroborate can be used:
- To prepare amino esters by reacting with lactams followed by hydrolysis.
- In the preparation of substituted imidazolines from aziridines and nitriles via [3+2]-cycloaddition reaction.
- For the N-alkylation of a series of N-arylsulfonyl-α-amino acid methyl esters having variable substituents at 4th position of the sulfonamide aromatic ring.
Other Notes
Powerful ethylating agent; Esterification of acids; Modifies carboxyl residues in proteins
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Dam. 1 - Skin Corr. 1B
Supplementary Hazards
Storage Class Code
8A - Combustible, corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
The identification of aspartic acid residue 52 as being critical to lysozyme activity.
S M Parsons et al.
Biochemistry, 8(10), 4199-4205 (1969-10-01)
M.J. Diem et al.
The Journal of Organic Chemistry, 42, 1801-1801 (1977)
D.J. Raber et al.
The Journal of Organic Chemistry, 44, 1149-1149 (1979)
N-Alkylation of N-arylsulfonyl-α-amino acid methyl esters by trialkyloxonium tetrafluoroborates
De Marco R, et al.
Tetrahedron, 67(50), 9708-9714 (2011)
Synthesis of substituted imidazolines via [3+ 2]-cycloaddition of aziridines with nitriles
Prasad BAB, et al.
Tetrahedron Letters, 45(6), 1137-1141 (2004)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service