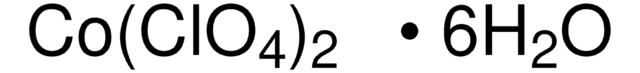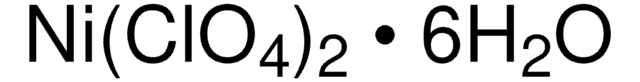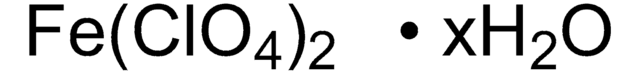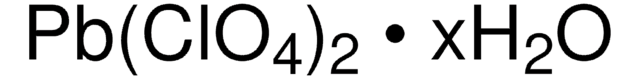215392
Copper(II) perchlorate hexahydrate
98%
Synonyme(s) :
Cupric perchlorate hexahydrate
About This Item
Produits recommandés
Pureté
98%
Forme
crystalline
Pertinence de la réaction
reagent type: oxidant
Densité
2.225 g/mL at 25 °C (lit.)
Application(s)
battery manufacturing
Chaîne SMILES
O.O.O.O.O.O.[Cu++].[O-]Cl(=O)(=O)=O.[O-]Cl(=O)(=O)=O
InChI
1S/2ClHO4.Cu.6H2O/c2*2-1(3,4)5;;;;;;;/h2*(H,2,3,4,5);;6*1H2/q;;+2;;;;;;/p-2
Clé InChI
NHELIHXBJRANPL-UHFFFAOYSA-L
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Catégories apparentées
Description générale
Application
- Methanol[1-(methoxymethanimidoyl)-2-(pyridin-2-ylmethyl)guanidine]bis(perchlorato)copper(II).: This study reports the synthesis and crystal structure of a new copper(II) complex with potential applications in materials science and catalysis. The research focuses on the structural characterization of the complex, which is crucial for understanding its reactivity and potential uses in various chemical processes (Meenongwa et al., 2012).
- Oxidative cleavage of DNA by a dipyridoquinoxaline copper(II) complex in the presence of ascorbic acid.: This research explores the catalytic activity of a copper(II) complex in oxidative DNA cleavage. The findings demonstrate the complex′s potential application in biochemical studies and the development of new catalytic processes for DNA manipulation and analysis (Santra et al., 2002).
Mention d'avertissement
Danger
Mentions de danger
Conseils de prudence
Classification des risques
Eye Irrit. 2 - Ox. Sol. 2 - Skin Irrit. 2 - STOT SE 3
Organes cibles
Respiratory system
Code de la classe de stockage
5.1A - Strongly oxidizing hazardous materials
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Équipement de protection individuelle
Eyeshields, Gloves, type P3 (EN 143) respirator cartridges
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique