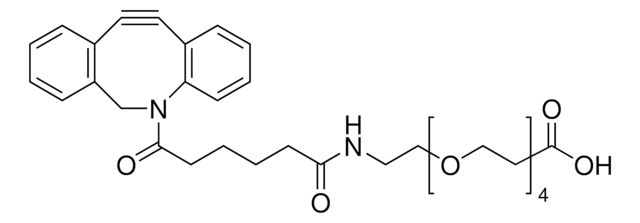
744867
(1R,8S,9s)-Bicyclo[6.1.0]non-4-yn-9-ylmethyl N-succinimidyl carbonate
for Copper-free Click Chemistry
Sinónimos:
N-[((1R,8S,9s)-Bicyclo[6.1.0]non-4-yn-9-yl)methyloxycarbonyloxy]succinimide, BCN-NHS, BCN-succinimidyl ester
About This Item
Productos recomendados
form
powder
composition
carbon content, 61.85%
hydrogen content, 5.88%
nitrogen content, 4.81%
reaction suitability
reaction type: click chemistry
reagent type: cross-linking reagent
functional group
NHS ester
storage temp.
−20°C
SMILES string
[H][C@@]12CCC#CCC[C@]1([H])[C@@H]2COC(ON3C(CCC3=O)=O)=O
InChI
1S/C15H17NO5/c17-13-7-8-14(18)16(13)21-15(19)20-9-12-10-5-3-1-2-4-6-11(10)12/h10-12H,3-9H2/t10-,11+,12-
InChI key
SKTDJYHCSCYLQU-ZSBIGDGJSA-N
Application
It may also be used to synthesize bicyclononyne functionalized poly(ethylene glycol) polymer coatings with anti-fouling properties towards protein adhesion and cell adhesion for supramolecular ureidopyrimidinone (UPy) based materials.
Packaging
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Artículos
Copper-free click chemistry is an alternative approach to click chemistry that proceeds at a lower activation barrier and is free of cytotoxic transition metal catalysts.
Drug discovery process by utilizing chemistry reaction of Cu(I)-catalyzed Huisgen 1,3-dipolar cycloaddition of terminal alkynes with organoazides to yield 1,4-disubstituted 1,2,3-triazoles.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico
![(1R,8S,9s)-Bicyclo[6.1.0]non-4-yn-9-ylmethanol for Copper-free Click Chemistry](/deepweb/assets/sigmaaldrich/product/structures/171/632/0556139a-2db5-4678-a6ec-a26a693fd574/640/0556139a-2db5-4678-a6ec-a26a693fd574.png)

![N-[(1R,8S,9s)-Bicyclo[6.1.0]non-4-yn-9-ylmethyloxycarbonyl]-1,8-diamino-3,6-dioxaoctane for Copper-free Click Chemistry](/deepweb/assets/sigmaaldrich/product/structures/294/853/c5e47d84-5aee-4797-aa24-604f291171cc/640/c5e47d84-5aee-4797-aa24-604f291171cc.png)