171778
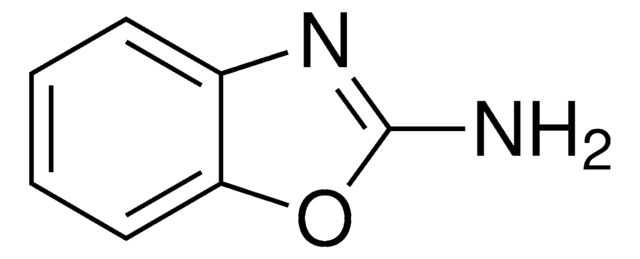
2-Aminobenzimidazole
97%
Sinónimos:
2-Benzimidazolamine
About This Item
Productos recomendados
assay
97%
form
solid
mp
226-230 °C (lit.)
SMILES string
Nc1nc2ccccc2[nH]1
InChI
1S/C7H7N3/c8-7-9-5-3-1-2-4-6(5)10-7/h1-4H,(H3,8,9,10)
InChI key
JWYUFVNJZUSCSM-UHFFFAOYSA-N
Gene Information
human ... PLAU(5328)
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
Application
signalword
Warning
hcodes
Hazard Classifications
Acute Tox. 4 Oral
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
ppe
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico