123617
Benzo[h]quinoline
97%
Sinónimos:
1-Naphthoquinoline, 7,8-Benzoquinoline
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula empírica (notación de Hill):
C13H9N
Número de CAS:
Peso molecular:
179.22
Beilstein:
120249
Número CE:
Número MDL:
Código UNSPSC:
12352100
ID de la sustancia en PubChem:
NACRES:
NA.22
Productos recomendados
Nivel de calidad
Ensayo
97%
Formulario
solid
bp
338 °C/719 mmHg (lit.)
mp
48-50 °C (lit.)
cadena SMILES
c1ccc2c(c1)ccc3cccnc23
InChI
1S/C13H9N/c1-2-6-12-10(4-1)7-8-11-5-3-9-14-13(11)12/h1-9H
Clave InChI
WZJYKHNJTSNBHV-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
Aplicación
Benzo[h]quinoline was used to study the mutagenic activities of benzo[f]quinoline, benzo[h]quinolone and a number of their derivatives in strain TA 100 of Salmonella typhimurium. It was used in determination of nitrogen-containing polynuclear aromatic hydrocarbons in the gaseous products of the thermal degradation of polymers by HPLC- fluorescence detection. It was used as starting reagent for the synthesis of osmium and ruthenium complexes containing an N-heterocyclic carbene ligand.
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
235.4 °F - closed cup
Punto de inflamabilidad (°C)
113 °C - closed cup
Equipo de protección personal
Eyeshields, Gloves, type N95 (US)
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
M Wilhelm et al.
Journal of chromatography. A, 878(2), 171-181 (2000-06-24)
A method for the simultaneous determination of 22 nitrogen-containing polynuclear aromatic hydrocarbons (PAHs) (15 azaarenes and seven amino-PAHs) in the gaseous products of the thermal degradation of polymers is described. After desorption and clean-up using cation-exchange chromatography (PRS cartridge) the
E J LaVoie et al.
Japanese journal of cancer research : Gann, 78(2), 139-143 (1987-02-01)
The environmental occurrence and mutagenic activity of quinoline and benzoquinolines are well-documented. In this study, the relative carcinogenic activities of quinoline, benzo[f]quinoline, benzo[h]quinoline, and phenanthridine were evaluated in newborn mice. Mice were injected intraperitoneally on the first, eighth, and fifteenth
E J LaVoie et al.
Carcinogenesis, 4(9), 1133-1138 (1983-09-01)
Benzo[f]quinoline and benzo[h]quinoline are widespread environmental pollutants which have been found to be mutagenic. The metabolism of benzo[f]quinoline and benzo[h]quinoline was investigated using a liver homogenate from Aroclor-pretreated rats. The metabolites of benzo[f]quinoline which were identified were 7,8-dihydroxy-7,8-dihydrobenzo[f]quinoline, 9,10-dihydroxy-9,10-dihydrobenzo[f]quinoline, 7-hydroxybenzo[f]quinoline
E C Riesgo et al.
Inorganic chemistry, 40(14), 3413-3422 (2001-06-26)
The Friedländer condensation was employed to synthesize two series of 3,3'-polymethylene bridged ligands, L, based on 2-(2'-pyridyl)-benzo[h]quinoline and 2,2'-bibenzo[h]quinoline (BHQ) along with the fully aromatic naphtho[1,2-b]-1,10-phenanthroline. Complexes [Cu(L)(2)](+) were prepared as their perchlorate or hexafluorophosphate salts. The solution state structures
Hanumantharao Paritala et al.
Bioorganic & medicinal chemistry letters, 19(6), 1584-1587 (2009-02-27)
G-quadruplexes are unusual structures formed from guanine-rich sequences of nucleic acids. G-quadruplexes have been postulated to play important roles in a number of biological systems including gene regulation and the inhibition of enzyme function. Recently, our laboratory reported on the
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico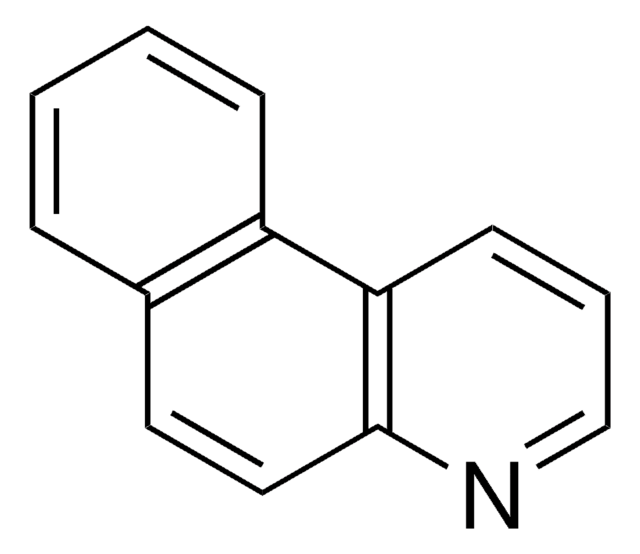










![Dibenz[c,h]acridine BCR®, certified reference material](/deepweb/assets/sigmaaldrich/product/structures/364/643/698df9fb-5b7d-467a-b47e-c8318e2ed298/640/698df9fb-5b7d-467a-b47e-c8318e2ed298.png)
![Benz[g]isoquinoline-5,10-dione 99%](/deepweb/assets/sigmaaldrich/product/structures/484/029/288c4a9d-19c2-4b51-82c1-f43b50ea05b0/640/288c4a9d-19c2-4b51-82c1-f43b50ea05b0.png)