323594
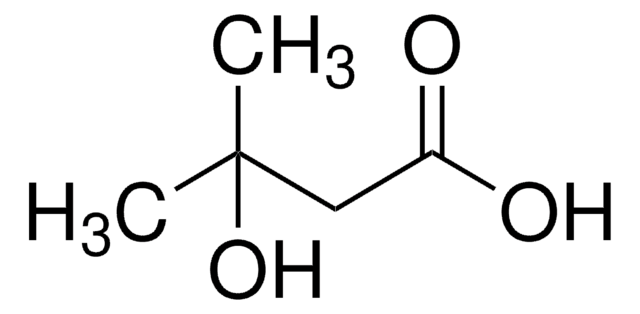
α-Hydroxyisobutyric acid
99%
Synonym(s):
alpha-Hydroxyisobutyric acid, 2-Hydroxy-2-methylpropionic acid, 2-Methyllactic acid
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
(CH3)2C(OH)COOH
CAS Number:
Molecular Weight:
104.10
Beilstein:
1744739
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
99%
form
solid
bp
84 °C/1.5 mmHg (lit.)
mp
76-80 °C (lit.)
functional group
carboxylic acid
hydroxyl
SMILES string
CC(C)(O)C(O)=O
InChI
1S/C4H8O3/c1-4(2,7)3(5)6/h7H,1-2H3,(H,5,6)
InChI key
BWLBGMIXKSTLSX-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
α-Hydroxyisobutyric acid is a versatile building block in organic synthesis. It serves as a precursor in the synthesis of compounds like isobutylene glycol and methacrylic acid
Application
α-Hydroxyisobutyric acid (HIBA) can be used as:
- A chelating agent to improve the separation of lanthanides and actinides by liquid chromatography.
- In the synthesis of room temperature–stabilized, pure, nanocrystalline β-NiMoO4.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Dam. 1 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Bimetallic single-source precursor for the synthesis of pure nanocrystalline room temperature-stabilized β-NiMoO4
Moneeb AM, et al.
Ceramics International, 42(1), 1366-1372 (2016)
Electrospray ionization mass spectrometric studies on uranyl complex with α-hydroxyisobutyric acid in water-methanol medium
Jaison PG, et al.
Rapid Communications in Mass Spectrometry, 27(10), 1105-1118 (2013)
From rotational resolved spectra to an extended increment system of planar moments allowing ad-hoc conformational identification ? Exemplification by the broadband microwave spectrum of ??- hydroxyisobutyric acid
P Buschmann, et al.
Journal of Molecular Structure, 1250, 131805-131805 (2022)
Thore Rohwerder et al.
Applied and environmental microbiology, 72(6), 4128-4135 (2006-06-06)
Fuel oxygenates such as methyl and ethyl tert-butyl ether (MTBE and ETBE, respectively) are degraded only by a limited number of bacterial strains. The aerobic pathway is generally thought to run via tert-butyl alcohol (TBA) and 2-hydroxyisobutyrate (2-HIBA), whereas further
Ana Carolina O Costa et al.
Journal of chromatography. A, 1171(1-2), 140-143 (2007-10-09)
The aim of this work was to develop a fast method using capillary electrophoresis for the determination of creatinine in human urine samples. The pH and constituents of the background electrolyte were selected by inspection of effective mobility of creatinine
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service