497401
(R)-(+)-2-Methyl-2-propanesulfinamide
98%
Synonym(s):
(R)-2-methyl-2-propanesulfinamide, (R)-2-methylpropane-2-sulfinamide, (R)-tert-butanesulfinamide, (R)-tert-butylsulfinamide
Sign Into View Organizational & Contract Pricing
All Photos(4)
About This Item
Linear Formula:
(CH3)3CS(O)NH2
CAS Number:
Molecular Weight:
121.20
MDL number:
UNSPSC Code:
12352111
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
optical activity
[α]20/D +4°, c = 1.0242 in chloroform stab. with amylenes
mp
103-107 °C (lit.)
storage temp.
2-8°C
SMILES string
CC(C)(C)S(N)=O
InChI
1S/C4H11NOS/c1-4(2,3)7(5)6/h5H2,1-3H3/t7-/m1/s1
InChI key
CESUXLKAADQNTB-SSDOTTSWSA-N
Related Categories
General description
(R)-(+)-2-Methyl-2-propanesulfinamide is a chiral auxiliary used in the condensation of the aldehyde.
Application
(R)-(+)-2-Methyl-2-propanesulfinamide may be used to prepare N-(1-cyclohexylmethylidene)-2-methylpropane-2-sulfinamide via copper mediated condensation with cyclohexane carboxaldehyde. It may also be used to prepare (20E)-N-[t-butyl-(R)-sulfinyl]-3β-(t-butyldimethylsilyloxy)-pregn-5-en-20-imine, an intermediate for the development of androgen receptor antagonists.
Can be readily transformed into P,N-sulfinyl imine ligands through condensation with aldehydes and ketones, which can undergo iridium-catalyzed asymmetric hydrogenation of olefins.
Preparation of ß-chloro sulfinamides in a synthesis of chiral azridines. Also used to prepare an organocatalyst for enantioselective reduction of imines.
Useful reagent for synthesizing chiral amines.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Bram Denolf et al.
Organic letters, 8(14), 3129-3132 (2006-06-30)
[reaction: see text] Reaction of chiral alpha-chloro tert-butanesulfinyl aldimines with Grignard reagents efficiently afforded beta-chloro N-sulfinamides in high diastereomeric excess. The latter compounds were cyclized toward the corresponding chiral aziridines in a high-yielding one-pot reaction or after separate treatment with
20-Aminosteroids as a novel class of selective and complete androgen receptor antagonists and inhibitors of prostate cancer cell growth.
Fousteris MA, et al.
Bioorganic & Medicinal Chemistry, 18(19), 6960-6969 (2010)
Jeffrey P McMahon et al.
Organic letters, 6(10), 1645-1647 (2004-05-07)
Addition of alkyl or aryl Grignard reagents to N-sulfinyl imines derived from 3- and 4-substituted cyclohexanones proceeds with good yields and with excellent diasteroselectivity. The selectivity of the reaction is controlled by the ring substituent rather than the sulfinyl group
Asymmetric synthesis of a, a-difluoro-?-amino acid derivatives from enantiomerically pure N-tert-butylsulfinimines.
Staas, DD, et al.
The Journal of Organic Chemistry, 67(23), 8276-8279 (2002)
Dong Pei et al.
Organic letters, 8(25), 5913-5915 (2006-12-01)
Easily accessible chiral sulfinamide 2 has been developed as the first highly efficient and enantioselective organocatalyst relying solely on a chiral sulfur center for stereochemical induction. In the presence of 20 mol % of 2, a broad range of N-aryl
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service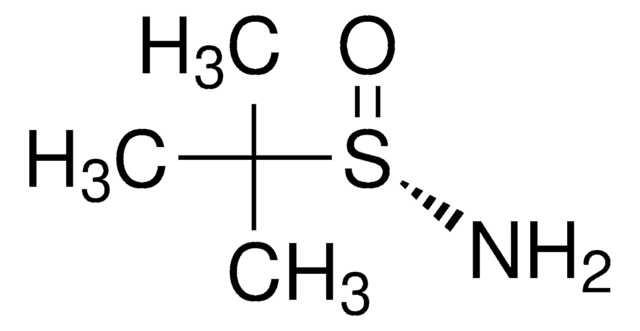








![(R)-N-[(1R,2R)-2-(3-(3,5-Bis(trifluoromethyl)phenyl)ureido)cyclohexyl]-tert-butyl-sulfinamide 96%](/deepweb/assets/sigmaaldrich/product/structures/389/070/18847164-c6a7-4b4e-abcb-2dbc22493a2d/640/18847164-c6a7-4b4e-abcb-2dbc22493a2d.png)