1275009
USP
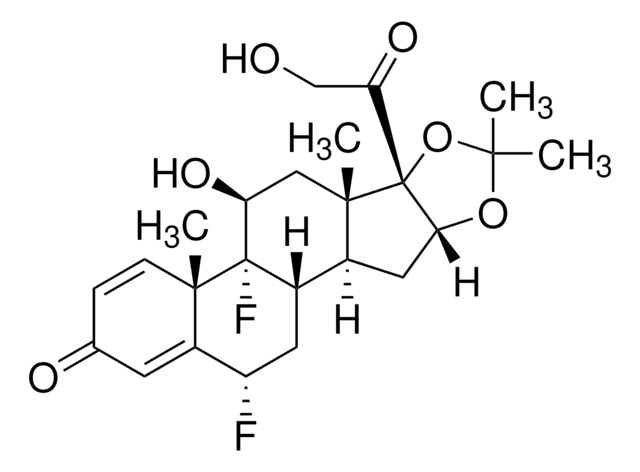
Fluocinolone acetonide
United States Pharmacopeia (USP) Reference Standard
Synonym(s):
Fluocinolone acetonide, 6α,9α-Difluoro-11β,16α,17α,21-tetrahydroxy-1,4-pregnadiene-3,20-dione, 6α,9α-Difluoro-16α-hydroxyprednisolone 16,17-acetonide, 6α-Fluorotriamcinolone acetonide, Sinalar, Synandone
About This Item
Recommended Products
grade
pharmaceutical primary standard
API family
fluocinolone
manufacturer/tradename
USP
mp
267-269 °C (lit.)
application(s)
pharmaceutical (small molecule)
format
neat
SMILES string
CC1(C)O[C@@H]2C[C@H]3[C@@H]4C[C@H](F)C5=CC(=O)C=C[C@]5(C)[C@@]4(F)[C@@H](O)C[C@]3(C)[C@@]2(O1)C(=O)CO
InChI
1S/C24H30F2O6/c1-20(2)31-19-9-13-14-8-16(25)15-7-12(28)5-6-21(15,3)23(14,26)17(29)10-22(13,4)24(19,32-20)18(30)11-27/h5-7,13-14,16-17,19,27,29H,8-11H2,1-4H3/t13-,14-,16-,17-,19+,21-,22-,23-,24+/m0/s1
InChI key
FEBLZLNTKCEFIT-VSXGLTOVSA-N
Gene Information
human ... NR3C1(2908)
Looking for similar products? Visit Product Comparison Guide
General description
Application
- Fluocinolone Acetonide Cream
- Fluocinolone Acetonide Ointment
- Fluocinolone Acetonide Topical Solution
- Neomycin Sulfate and Fluocinolone Acetonide Cream
Analysis Note
Other Notes
related product
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service