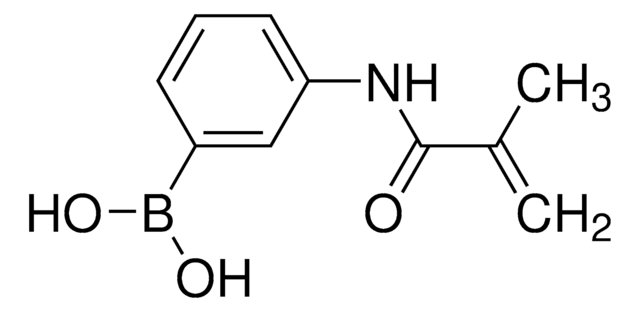
771465
3-(Acrylamido)phenylboronic acid
98%
Synonym(s):
3-(Propenamido)phenylboronic acid, N-Acryloyl-3-aminophenylboronic acid, Boronic acid acrylamide, Phenylboronate acrylamide
About This Item
Recommended Products
Assay
98%
form
powder
mp
129-146 °C
storage temp.
2-8°C
SMILES string
OB(O)c1cccc(NC(=O)C=C)c1
InChI
1S/C9H10BNO3/c1-2-9(12)11-8-5-3-4-7(6-8)10(13)14/h2-6,13-14H,1H2,(H,11,12)
InChI key
ULVXDHIJOKEBMW-UHFFFAOYSA-N
General description
Application
- As a monomer to synthesize poly(methacrylic acid)-co-3-(acrylamido)phenylboronic acid (PMAA-co-AAPBA) copolymer as a supramolecular receptor for biosensor applications. AAPBA helps to enhance the water solubility and binding affinity of the copolymer. This copolymer is utilized for carbohydrate sensing in an aqueous medium.
- As a monomer to prepare poly(3-Acrylamidophenyl boronic acid-b-diethylene glycol dimethacrylate) for the fabrication of glucose-sensitive nanoparticles for insulin delivery. The specific interactions of AAPBA with the diol moiety present in glucose molecules induce glucose responsiveness into the block copolymer.
- As a monomer and cross-linker to synthesize self-healing composite hydrogels for tissue engineering and drug delivery systems. They can mimic the properties of natural tissues and provide a suitable environment for cell growth. AAPBA polymerizes with acrylamide and simultaneously interacts with cis-diol of hydroxypropyl guar gum (HPG) to facilitate the formation of hydrogel with good mechanical strength and fast self-healing properties.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service