Select a Size
| Pack Size | SKU | Availability | Price |
|---|---|---|---|
| 25 mg | Please contact Customer Service for Availability | CZK 5,340.00 | |
| 100 mg | Please contact Customer Service for Availability | CZK 17,000.00 | |
| 500 mg | Please contact Customer Service for Availability | CZK 62,200.00 |
About This Item
form
solid
Quality Level
optical activity
[α]28/D +3.8°, c = 0.9 in methanol(lit.)
storage condition
desiccated
color
white
solubility
DMSO: >20 mg/mL, H2O: 26 mg/mL (Solutions may be stored for several weeks at 4 °C.)
SMILES string
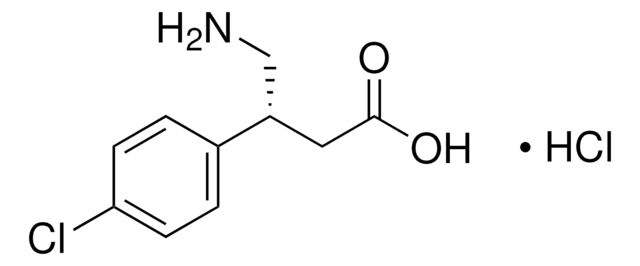
Cl[H].NC[C@H](CC(O)=O)c1ccc(Cl)cc1
InChI
1S/C10H12ClNO2.ClH/c11-9-3-1-7(2-4-9)8(6-12)5-10(13)14;/h1-4,8H,5-6,12H2,(H,13,14);1H/t8-;/m0./s1
InChI key
WMNUVYYLMCMHLU-QRPNPIFTSA-N
Gene Information
human ... GABBR1(2550)
mouse ... GABBR1(54393)
rat ... GABBR1(81657)
1 of 1
This Item | |||
|---|---|---|---|
| Quality Level 100 | Quality Level 100 | Quality Level 100 | Quality Level 200 |
| form solid | form solid | form solid | form crystalline powder |
| Gene Information human ... GABBR1(2550) | Gene Information human ... GABBR1(2550), GABBR2(9568) | Gene Information human ... GABBR1(2550), GABBR2(9568) | Gene Information human ... GABRA1(2554), GABRA2(2555), GABRA3(2556), GABRA4(2557), GABRA5(2558), GABRA6(2559), GABRB1(2560), GABRB2(2561), GABRB3(2562), SERPINA6(866) |
| storage condition desiccated | storage condition - | storage condition - | storage condition - |
| optical activity [α]28/D +3.8°, c = 0.9 in methanol(lit.) | optical activity - | optical activity - | optical activity - |
| color white | color white | color white to very faintly yellow | color - |
Application
Biochem/physiol Actions
Other Notes
Storage Class
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Protocols
Qualitative Thin Layer Chromatography Analysis of Flavonoids and Quantification of Terpene Lactones in Ginkgo Biloba Extracts and Tablets
Global Trade Item Number
| SKU | GTIN |
|---|---|
| G013-100MG | 04061833623213 |
| G013-25MG | 04061833623220 |
Active Filters
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service