66940
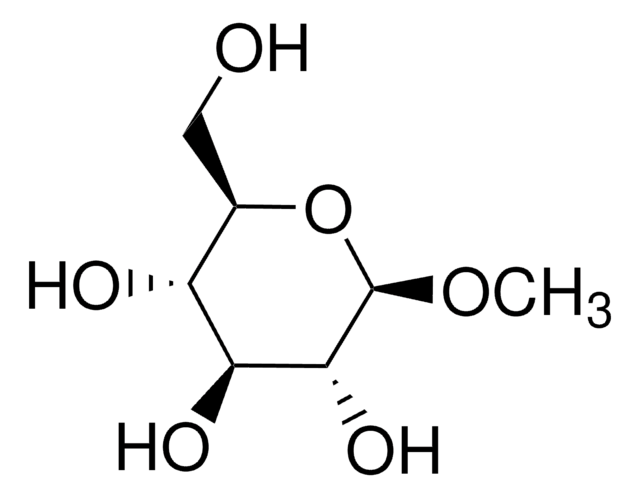
Methyl α-D-glucopyranoside
≥99.0%, suitable for microbiology
Synonym(s):
alpha-Methyl D-glucose ether, Methyl glucose, Methyl alpha-D-glucoside, Methyl glucopyranoside, Methyl alpha-D-glucose, Methyl pyranoside, Methyl α-D-glucoside
About This Item
Recommended Products
Quality Level
assay
≥99.0% (sum of enantiomers, HPLC)
≥99.0%
form
crystalline powder
optical activity
[α]20/D +157±3°, c = 10% in H2O
packaging
pkg of 100 g
storage condition
(Keep container tightly closed in a dry and well-ventilated place.)
color
colorless
mp
165-169 °C
application(s)
microbiology
SMILES string
CO[C@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O
InChI
1S/C7H14O6/c1-12-7-6(11)5(10)4(9)3(2-8)13-7/h3-11H,2H2,1H3/t3-,4-,5+,6-,7+/m1/s1
InChI key
HOVAGTYPODGVJG-ZFYZTMLRSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
Storage Class
11 - Combustible Solids
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Culture media provides a habitat with suitable nutrients, energy sources, and certain environmental conditions for the growth of microorganisms. The components of the culture media range from simple sugars to peptones, salts, antibiotics, and complex indicators.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service