All Photos(1)
About This Item
Empirical Formula (Hill Notation):
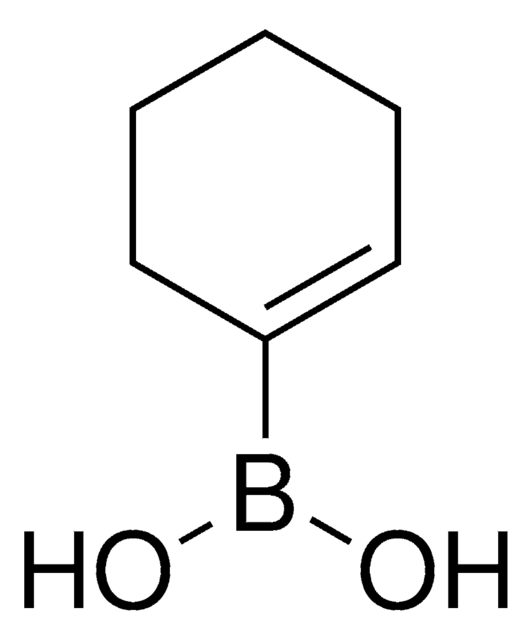
C6H13BO2
CAS Number:
Molecular Weight:
127.98
MDL number:
UNSPSC Code:
12352103
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
assay
≥95%
form
powder
SMILES string
OB(O)C1CCCCC1
InChI
1S/C6H13BO2/c8-7(9)6-4-2-1-3-5-6/h6,8-9H,1-5H2
InChI key
XDRVAZAFNWDVOE-UHFFFAOYSA-N
Related Categories
Application
Reactant involved in:
- HF-free synthesis of tetrabutylammonium trifluoroborates
- Cross-coupling with aromatic amines
- Suzuki cross-coupling reactions
- Arylation and alkylation of diphenylisoxazole
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Mirei Shou et al.
Chirality, 27(7), 417-421 (2015-05-23)
Three aldohexoses, glucose, galactose, and mannose, and three aldopentoses, arabinose, xylose, and ribose, were derivatized with L-tryptophanamide (L-TrpNH2 ) under alkaline conditions. Using a basic mobile phase (pH 9.2), the three aldohexoses or the three aldopentoses were simultaneously enantioseparated, respectively, but
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service