All Photos(1)
About This Item
Linear Formula:
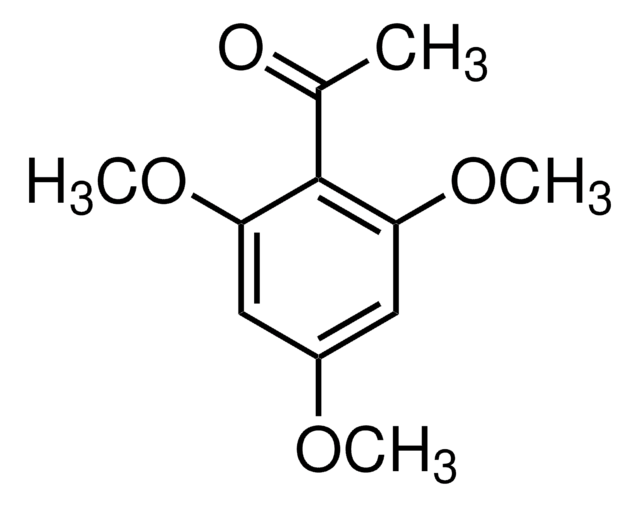
(CH3O)2C6H3COCH3
CAS Number:
Molecular Weight:
180.20
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
assay
98%
form
solid
bp
135-136 °C/2 mmHg (lit.)
mp
68-70 °C (lit.)
functional group
ketone
SMILES string
COc1cccc(OC)c1C(C)=O
InChI
1S/C10H12O3/c1-7(11)10-8(12-2)5-4-6-9(10)13-3/h4-6H,1-3H3
InChI key
XEUGKOFTNAYMMX-UHFFFAOYSA-N
General description
2,6-Dihydroxyacetophenone, its mono- and di-methyl ethers are inhibitors of hepatic mixed function oxidases. Metabolism of 2′,6′-dimethoxyacetophenone was studied.
Application
2′,6′-Dimethoxyacetophenone was used in preparation of 4-fluororesorcinol.
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Facile preparations of 4-fluororesorcinol.
Belanger PC, et al.
Canadian Journal of Chemistry, 66(6), 1479-1482 (1988)
A Bobik et al.
Xenobiotica; the fate of foreign compounds in biological systems, 5(2), 65-72 (1975-02-01)
1. 2,6-Dihydroxyacetophenone, its mono- and di-methyl ethers are inhibitors of hepatic mixed function oxidases. The dimethyl ether is a competitive inhibitor of aminopyrine demethylase with the others displaying mixed kinetics. The metabolism of all three ketones has been studied. 2.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service