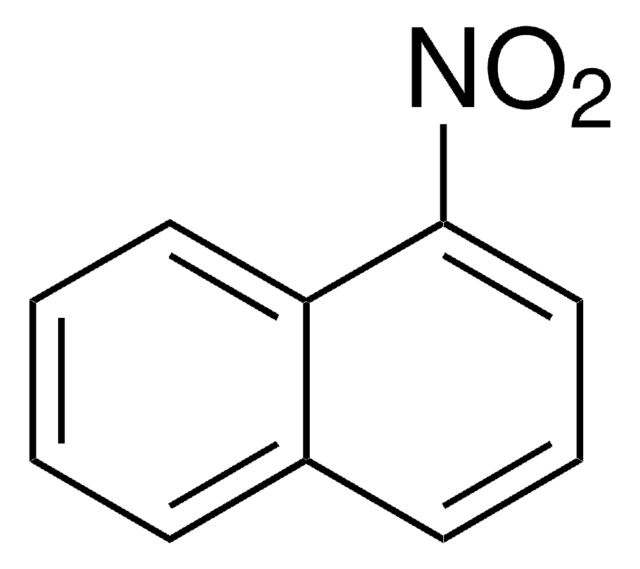
103543
4-Nitroanisole
97%
Synonym(s):
1-Methoxy-4-nitrobenzene
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
O2NC6H4OCH3
CAS Number:
Molecular Weight:
153.14
Beilstein/REAXYS Number:
1865361
EC Number:
MDL number:
UNSPSC Code:
12352100
eCl@ss:
39032065
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
assay
97%
form
solid
density
1.233 g/mL at 25 °C (lit.)
functional group
nitro
SMILES string
COc1ccc(cc1)[N+]([O-])=O
InChI
1S/C7H7NO3/c1-11-7-4-2-6(3-5-7)8(9)10/h2-5H,1H3
InChI key
BNUHAJGCKIQFGE-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
4-Nitroanisole undergoes photochemical nucleophilic aromatic substitution by hydroxide ion to form 4-methoxyphenol and 4-nitrophenol.
Application
4-Nitroanisole was used as probe to determine Π* of Kamlet-Taft solvent parameters and high pressure and supercritical water in the temperature range of 16-420 °C. 4-Nitroanisole was used as carbon and energy supplement for the isolation of Rhodococcus strains.
Biochem/physiol Actions
4-Nitroanisole is O-demethylated to 4-nitrophenol by human liver microsomes.
signalword
Warning
hcodes
Hazard Classifications
Aquatic Chronic 3 - Carc. 2
Storage Class
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
wgk_germany
WGK 1
flash_point_f
266.0 °F - closed cup
flash_point_c
130 °C - closed cup
ppe
Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Vadim Samoilov et al.
Molecules (Basel, Switzerland), 25(7) (2020-04-15)
In this study approaches for chemical conversions of the renewable compounds 1,2-propanediol (1,2-PD) and 2,3-butanediol (2,3-BD) that yield the corresponding cyclic ketals and glycol ethers have been investigated experimentally. The characterization of the obtained products as potential green solvents and
H V Gelboin et al.
Biochemical pharmacology, 50(11), 1841-1850 (1995-11-27)
Cytochromes P450 3A3/4 are inordinately important P450 enzymes catalyzing the metabolism of a large variety of clinically useful drugs, steroids, and carcinogens. Two monoclonal antibodies, MAb 3-29-9 and MAb 275-1-2, were prepared to human P450 3A4 from mice immunized with
H V Gelboin et al.
Chemical research in toxicology, 9(6), 1023-1030 (1996-09-01)
A panel of 17 hybridomas producing (MAbs) against human cytochrome P450 2E1 (h2E1) was generated by immunizing mice with baculovirus-expressed h2E1. All 17 hybridoma clones gave positive ELISA or immunoblots with either baculovirus-or vaccinia virus-expressed h2E1. Two of the latter
S F Simoni et al.
Journal of contaminant hydrology, 50(1-2), 99-120 (2001-07-28)
Microbial degradation rates in the subsurface are not only limited by the physiological capacity of the organisms, but also by inefficient supply of nutrients to the microbes. Although mass transfer limitation of biodegradation in the subsurface has been postulated for
V V Shumyantseva et al.
Biochemistry and molecular biology international, 45(1), 171-179 (1998-06-23)
Semisynthetic flavocytochromes, obtained by covalent binding of riboflavin with cytochromes P450 2B4, were able to catalyze the H2O2-mediated reactions of aniline p-hydroxylation, aminopyrine N-demethylation and p-nitroanizole' O-dealkylation. The rates of the flavocytochrome-catalyzed, H2O2-supported reactions far exceeded those of the appropriate
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service