U4375
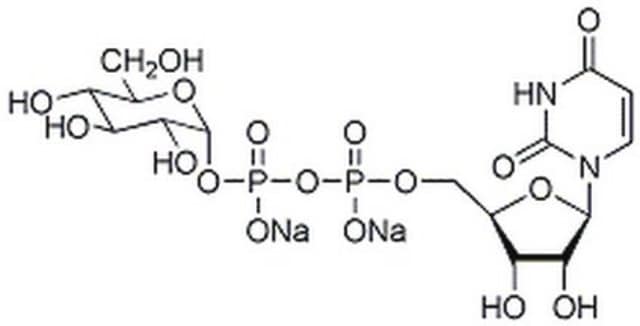
Uridine 5′-diphospho-N-acetylglucosamine sodium salt
≥98%
Synonym(s):
UDP-GlcNAc, UDP-N-acetylglucosamine, UDPAG
Select a Size
Select a Size
About This Item
Recommended Products
biological source
bakers yeast
Quality Level
assay
≥98%
form
powder
storage temp.
−20°C
SMILES string
[Na+].[Na+].CC(=O)N[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1OP([O-])(=O)OP([O-])(=O)OC[C@H]2O[C@H]([C@H](O)[C@@H]2O)N3C=CC(=O)NC3=O
InChI
1S/C17H27N3O17P2.2Na/c1-6(22)18-10-13(26)11(24)7(4-21)35-16(10)36-39(31,32)37-38(29,30)33-5-8-12(25)14(27)15(34-8)20-3-2-9(23)19-17(20)28;;/h2-3,7-8,10-16,21,24-27H,4-5H2,1H3,(H,18,22)(H,29,30)(H,31,32)(H,19,23,28);;/q;2*+1/p-2/t7-,8-,10-,11-,12-,13-,14-,15-,16-;;/m1../s1
InChI key
HXWKMJZFIJNGES-YZVFIFBQSA-L
Related Categories
General description
Application
- as a component of reaction cocktail in endoplasmic reticulum to Golgi transport assay[4]
- as a reference standard for the quantification of UDP-GlcNAc in liver tissues using high-performance liquid chromatography (HPLC)[5]
- in testing the glycosylation activity of O-GlcNAc transferase (OGT) against peptide substrate[6]
Biochem/physiol Actions
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Explore tools for glycosyltransferase synthesis and modification of glycans, such as glycosyltransferases and nucleotide sugar donors.
LC-MS/MS method quantifies similar polar nucleotide activated sugars using Supel™ Carbon LC column for simultaneous analysis.
Enzymatic glycosyltransferase specificity challenges the one enzyme-one linkage concept.
Understand sialic acid structure, function, signaling, and modifications. Easily find products for sialic acid research.
Active Filters
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service