28266
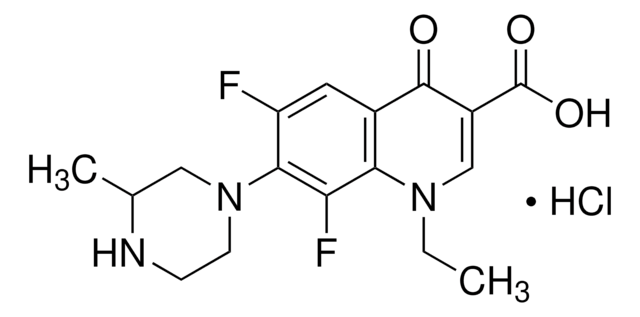
Levofloxacin
98.0-102.0% anhydrous basis (HPLC)
Synonym(s):
Levofloxacin Anhydrous, (−)-Ofloxacin
About This Item
Recommended Products
biological source
synthetic
Quality Level
assay
98.0-102.0% anhydrous basis (HPLC)
form
powder or crystals
color
light yellow-white to yellow-white
antibiotic activity spectrum
Gram-negative bacteria
Gram-positive bacteria
mode of action
enzyme | inhibits
SMILES string
OC(C(C1=O)=CN2C(C1=CC(F)=C3N4CCN(C)CC4)=C3OC[C@@H]2C)=O
InChI
1S/C18H20FN3O4/c1-10-9-26-17-14-11(16(23)12(18(24)25)8-22(10)14)7-13(19)15(17)21-5-3-20(2)4-6-21/h7-8,10H,3-6,9H2,1-2H3,(H,24,25)/t10-/m0/s1
InChI key
GSDSWSVVBLHKDQ-JTQLQIEISA-N
Gene Information
human ... KCNH1(3756)
Looking for similar products? Visit Product Comparison Guide
Related Categories
application
Biochem/physiol Actions
Packaging
Other Notes
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Resp. Sens. 1A - Skin Sens. 1A
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Quinolones are a key group of antibiotics that interfere with DNA synthesis by inhibiting topoisomerase, most frequently topoisomerase II (DNA gyrase), an enzyme involved in DNA replication.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service