Sign In to View Organizational & Contract Pricing.
Select a Size
50 G
$71.70
250 G
$188.00
500 G
$222.00
About This Item
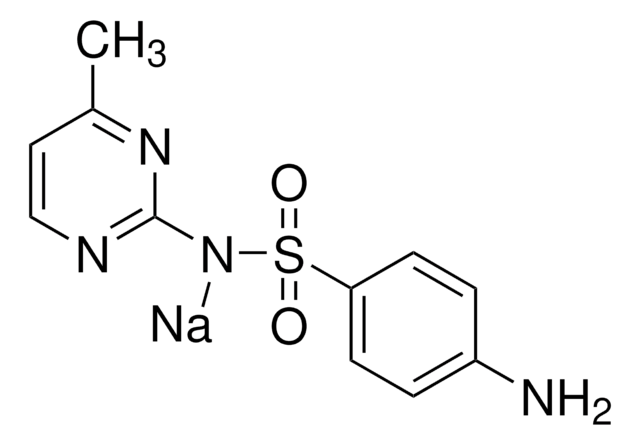
Empirical Formula (Hill Notation):
C11H12N4O2S
CAS Number:
Molecular Weight:
264.30
UNSPSC Code:
12352100
NACRES:
NA.22
PubChem Substance ID:
EC Number:
204-866-2
Beilstein/REAXYS Number:
249133
MDL number:
Assay:
≥99.0%
Skip To
InChI key
QPPBRPIAZZHUNT-UHFFFAOYSA-N
InChI
1S/C11H12N4O2S/c1-8-6-7-13-11(14-8)15-18(16,17)10-4-2-9(12)3-5-10/h2-7H,12H2,1H3,(H,13,14,15)
SMILES string
Cc1ccnc(NS(=O)(=O)c2ccc(N)cc2)n1
product line
ReagentPlus®
assay
≥99.0%
Quality Level
Looking for similar products? Visit Product Comparison Guide
Related Categories
1 of 4
This Item | |||
|---|---|---|---|
| assay ≥99.0% | assay - | assay - | assay 99.0-101.0% |
| Quality Level 200 | Quality Level 100 | Quality Level 200 | Quality Level 200 |
| product line ReagentPlus® | product line - | product line VETRANAL® | product line - |
Legal Information
ReagentPlus is a registered trademark of Merck KGaA, Darmstadt, Germany
signalword
Warning
hcodes
Hazard Classifications
Acute Tox. 4 Oral
Storage Class
11 - Combustible Solids
wgk
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Florian M Koch et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 17(13), 3679-3692 (2011-03-03)
The first catalytic asymmetric synthesis of β-sultones is reported. This development has enabled a rapid access to a number of highly enantioenriched biologically interesting sulfonyl and sulfinyl compound classes, which makes use of the inherent ring strain of the four-membered
A practical sulfenylation of 2,5-diketopiperazines.
K C Nicolaou et al.
Angewandte Chemie (International ed. in English), 51(3), 728-732 (2011-12-14)
Yi Li et al.
International journal of pharmaceutics, 415(1-2), 110-118 (2011-06-08)
The ability to detect and quantify polymorphism of pharmaceuticals is critically important in ensuring that the formulated product delivers the desired therapeutic properties because different polymorphic forms of a drug exhibit different solubilities, stabilities and bioavailabilities. The purpose of this
Mehdi D Esrafili et al.
Journal of molecular graphics & modelling, 27(3), 326-331 (2008-07-08)
A density functional theory investigation was carried out to characterize (14)N electric field gradient tensors, EFG, in crystalline sulfamerazine and sulfathiazole. To include hydrogen-bonding effects in the calculations, the most probable interacting molecules with the target were considered as tetrameric
Jia Pan et al.
Chemical communications (Cambridge, England), 47(1), 352-354 (2010-08-24)
A phosphine-mediated one-step disulfide formation from S-nitrosothiols has been developed. This reaction can convert unstable S-nitrosothiols to stable disulfides via sulfenamide intermediates under very mild conditions. It has the potential to be used for the detection of S-nitrosothiols.
Active Filters
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service