Sign In to View Organizational & Contract Pricing.
Select a Size
Pricing and availability is not currently available.
About This Item
Empirical Formula (Hill Notation):
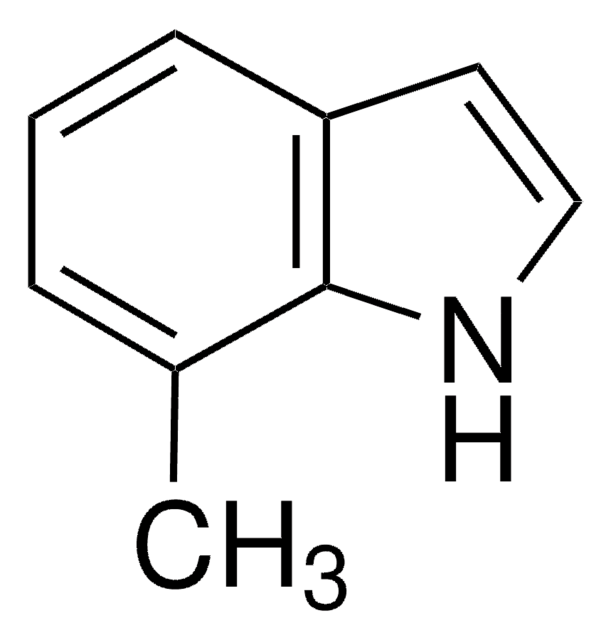
C9H9N
CAS Number:
Molecular Weight:
131.17
UNSPSC Code:
12352100
NACRES:
NA.22
PubChem Substance ID:
EC Number:
202-398-3
Beilstein/REAXYS Number:
109781
MDL number:
Assay:
98%
Skip To
Quality Level
assay
98%
bp
273 °C (lit.)
mp
57-59 °C (lit.)
SMILES string
Cc1cc2ccccc2[nH]1
InChI
1S/C9H9N/c1-7-6-8-4-2-3-5-9(8)10-7/h2-6,10H,1H3
InChI key
BHNHHSOHWZKFOX-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
1 of 4
This Item | |||
|---|---|---|---|
| assay 98% | assay 97% | assay 99% | assay 98% |
| Quality Level 200 | Quality Level 100 | Quality Level 100 | Quality Level 200 |
| mp 57-59 °C (lit.) | mp 80-84 °C (lit.) | mp 86-88 °C (lit.) | mp - |
| bp 273 °C (lit.) | bp 266 °C (lit.) | bp - | bp 114 °C/25 mmHg (lit.) |
Application
Reactant for:
- Regioselective synthesis of oxopyrrolidine analogs via iodine-catalyzed Markovnikov addition reaction
- Friedel-Crafts alkylation reactions
- Preparation of tryptophan dioxygenase inhibitors pyridyl-ethenyl-indoles as potential anticancer immunomodulators
- Preparation of plant-growth inhibitors
- Michael addition reactions
- Synthesis of cyclooxygenase-1 (COX-1)/cyclooxygenase-2 (COX-2) inhibitors
signalword
Warning
hcodes
Hazard Classifications
Acute Tox. 4 Oral
Storage Class
11 - Combustible Solids
wgk
WGK 3
flash_point_f
285.8 °F
flash_point_c
141 °C
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
[2-Methylindoles substituted in the 1st, 3d and 5th positions and the diffuse neuroendocrine APUD system].
K S Shadurskiĭ et al.
Farmakologiia i toksikologiia, 46(2), 115-120 (1983-03-01)
J M Gutteridge
The International journal of biochemistry, 14(7), 649-653 (1982-01-01)
1. The thiobarbituric acid (TBA) reaction, widely applied to the detection of autoxidation in polyunsaturated fatty acids, can be used to measure free-radical damage to amino acids, carbohydrates and nucleic acids. 2. In all of these systems malondialdehyde (MDA) is
V F Ximenes et al.
Archives of biochemistry and biophysics, 387(2), 173-179 (2001-05-24)
The indole moeity is present in many substances of biological occurrence. Its metabolism, in most cases, involves an oxidative pathway. This study reports the oxidation of a series of indole derivatives, including several of biological origin, catalyzed by horseradish peroxidase
Nevin Erciyes et al.
Journal of colloid and interface science, 278(1), 91-95 (2004-08-18)
The adsorption of indole and its 2-methyl derivative from aqueous solutions onto cobalt(II)-carboxylated diaminoethane sporopollenin (CDAE-sporopollenin) was studied using a fixed-bed column at 25+/-0.1 degrees C. Minicolumn adsorption studies showed that the breakthrough and the total adsorption capacities of CDAE-sporopollenin
T Bhattacharya et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 60(8-9), 1957-1966 (2004-07-14)
Electrochemical measurements by cyclic voltammetry predict the possibility of occurrence of photoinduced electron-transfer (PET) reactions between the ground state of 2-phenylindole (2PI) (electron donor) and the excited singlet of 9-cyanoanthracene (9CNA) molecule acting as an electron acceptor. However, 2PI should
Global Trade Item Number
| SKU | GTIN |
|---|---|
| M51407-100G | 04061838355058 |
| M51407-25G | 04061834059530 |
Active Filters
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service