All Photos(1)
About This Item
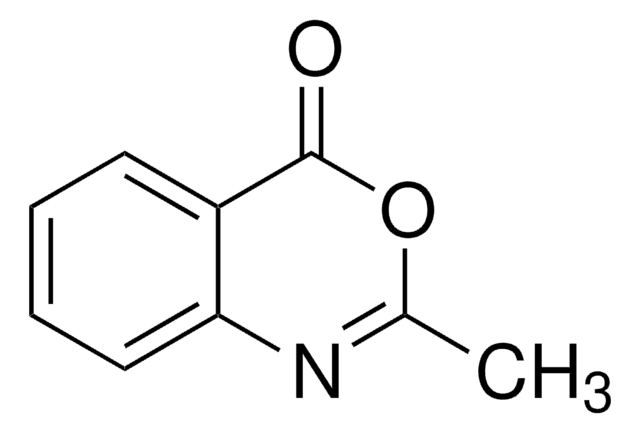
Empirical Formula (Hill Notation):
C9H9N3O
CAS Number:
Molecular Weight:
175.19
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
Recommended Products
assay
98%
form
solid
mp
149-152 °C (lit.)
SMILES string
CC1=Nc2ccccc2C(=O)N1N
InChI
1S/C9H9N3O/c1-6-11-8-5-3-2-4-7(8)9(13)12(6)10/h2-5H,10H2,1H3
InChI key
IQOUPYQSZBDNAW-UHFFFAOYSA-N
General description
3-Amino-2-methyl-4(3H)quinazolinone is a quinazoline derivative. 2-Methyl-3,1-benzoxazin-4-one undergoes condensation reaction with hydrazine hydrate to yield 3-amino-2-methyl-4(3H)quinazolinone. It undergoes condensation with various substituted aldehydes to afford Schiff′s bases.
Application
3-Amino-2-methyl-4(3H)quinazolinone may be used to synthesize:
- 2-methyl-3-[3′-aminophthalimido]-4(3H)-quinazolinone
- 2-alkyl-3-(methylamino)-4(3H)-quinazolinone
- 3-amino-2-chloromethyl-4(3H)-quinazolinone
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Disperse dyes based on 2-methyl-3-[3'-amino-phthalimido]-4 (3H)-quinazilinone.
Patel VH, et al.
J. Serb. Chem. Soc., 67(11), 719-726 (2002)
Lithiation of 2-Alkyl-3-amino-and 2-Alkyl-3-(methylamino)-4 (3 H)-quinazolinones1.
Smith K, et al.
The Journal of Organic Chemistry, 61(2), 656-661 (1996)
Dipolar Cycloaddition Reactions with Quinazolinones: A New Route for the Synthesis of Several Annelated Pyrrolo-and Pyridazinoquinazoline Derivatives.
Ghabrial SS and Gaber HM.
Molecules (Basel), 8(5), 401-410 (2003)
Kollur Shiva Prasad et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 81(1), 276-282 (2011-07-12)
Four Schiff base ligands and their corresponding organotin(IV) complexes have been synthesized and characterized by elemental analyses, IR, (1)H NMR, MS and thermal studies. The Schiff bases are obtained by the condensation of 3-amino-2-methyl-4(3H)-quinazolinone with different substituted aldehydes. The elemental
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service