436178
1-Ethylpiperidine hypophosphite
95%
Synonym(s):
1-Ethylpiperidinium phosphinate
About This Item
Recommended Products
assay
95%
form
solid
mp
40-43 °C (lit.)
storage temp.
2-8°C
SMILES string
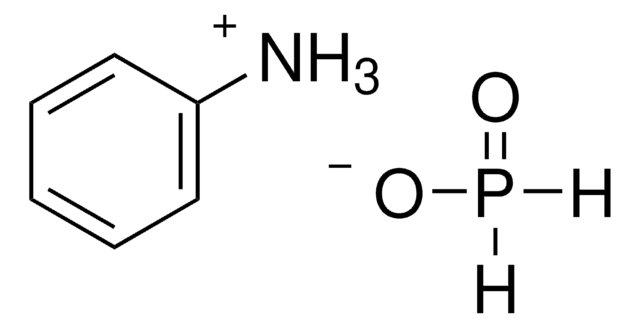
OPO.CCN1CCCCC1
InChI
1S/C7H15N.H3O2P/c1-2-8-6-4-3-5-7-8;1-3-2/h2-7H2,1H3;1-3H
InChI key
BNCZSWZTYPTERM-UHFFFAOYSA-N
General description
Application
- As water-soluble chain carrier for the efficient radical cyclization in water for various hydrophobic substrates.
- In the main radical-cyclisation step for the synthesis of the phytotoxic metabolite alboatrin and its epimer.
- Preparation of bifunctional pentafluorophenyl/2,4,6-trichlorophenyl sulfonates.
- Chemoselective radical reduction of the iodine atom in 1-deoxy-1-halo-1-iodo-alditols.
Synthesis of spiroketals via intramolecular iodoetherification
A facile radical cyclization diverted to a rearrangement-cyclization with base
Radical deoxygenation
Synthesis of allylic H-phosphinates
signalword
Warning
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Eye Irrit. 2 - Flam. Sol. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
4.1B - Flammable solid hazardous materials
wgk_germany
WGK 3
flash_point_f
132.8 °F - closed cup
flash_point_c
56 °C - closed cup
ppe
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service