Select a Size
$212.00
$672.00
$212.00
Available to ship TODAYDetails
About This Item
Skip To
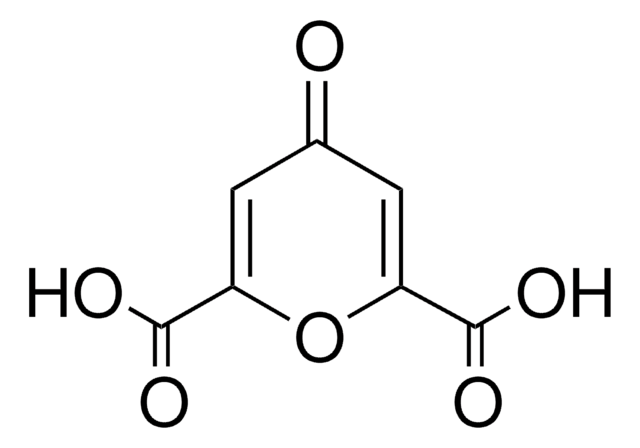
assay
≥95%
form
powder
mp
265 °C (dec.) (lit.)
functional group
carboxylic acid, ether, ketone
SMILES string
OC(=O)C1=CC(=O)C=C(O1)C(O)=O
InChI
1S/C7H4O6/c8-3-1-4(6(9)10)13-5(2-3)7(11)12/h1-2H,(H,9,10)(H,11,12)
InChI key
PBAYDYUZOSNJGU-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
1 of 4
This Item | |||
|---|---|---|---|
| assay ≥95% | assay - | assay ≥98.0% (HPLC) | assay ≥97.0% (dried material, T) |
| Quality Level 100 | Quality Level 100 | Quality Level - | Quality Level 200 |
| form powder | form powder | form powder | form powder |
| mp 265 °C (dec.) (lit.) | mp 265-270 °C | mp 265 °C (dec.) (lit.) | mp 267 °C (dec.) (lit.) |
| functional group carboxylic acid | functional group carboxylic acid | functional group - | functional group carboxylic acid |
General description
Application
Storage Class
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 382272-5G | 04061831962451 |
| 382272-25G | 04061833424742 |
Active Filters
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service