344508
Cyclopentene
96%
Synonym(s):
2,2-Dimethylacetyl chloride
About This Item
Recommended Products
vapor pressure
20.89 psi ( 55 °C)
6.11 psi ( 20 °C)
assay
96%
form
liquid
autoignition temp.
743 °F
refractive index
n20/D 1.421 (lit.)
bp
44-46 °C (lit.)
mp
−135 °C (lit.)
solubility
water: soluble 0.535 g/L at 25 °C
density
0.771 g/mL at 25 °C (lit.)
storage temp.
2-8°C
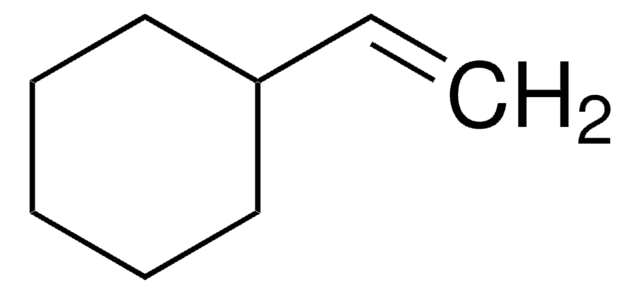
SMILES string
C1CC=CC1
InChI
1S/C5H8/c1-2-4-5-3-1/h1-2H,3-5H2
InChI key
LPIQUOYDBNQMRZ-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
recommended
signalword
Danger
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Oral - Asp. Tox. 1 - Flam. Liq. 2 - Skin Irrit. 2
Storage Class
3 - Flammable liquids
wgk_germany
WGK 2
flash_point_f
-29.9 °F - closed cup
flash_point_c
-34.4 °C - closed cup
ppe
Eyeshields, Faceshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service