About This Item
Skip To
assay
95%
form
liquid
impurities
2-5% 4-methoxy-3-buten-2-one
refractive index
n20/D 1.454 (lit.)
bp
68-69 °C/14 mmHg (lit.)
density
0.885 g/mL at 25 °C (lit.)
functional group
ether
storage temp.
2-8°C
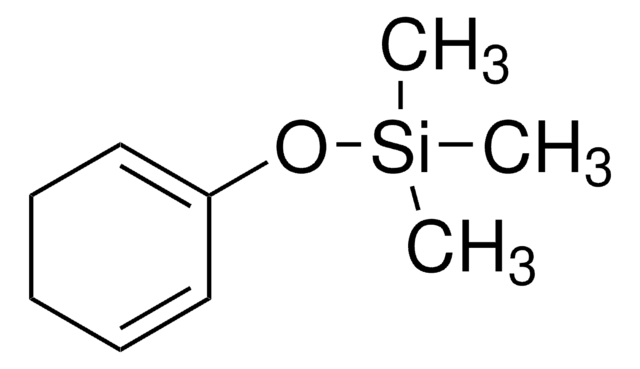
SMILES string
CO\C=C\C(=C)O[Si](C)(C)C
InChI
1S/C8H16O2Si/c1-8(6-7-9-2)10-11(3,4)5/h6-7H,1H2,2-5H3/b7-6+
InChI key
SHALBPKEGDBVKK-VOTSOKGWSA-N
Related Categories
1 of 4
This Item | |||
|---|---|---|---|
| assay 95% | assay 98% | assay 95% | assay 95% |
| Quality Level 100 | Quality Level 200 | Quality Level 100 | Quality Level 100 |
| bp 68-69 °C/14 mmHg (lit.) | bp 68-69 °C (lit.) | bp 134-136 °C/745 mmHg (lit.) | bp 65 °C/7 mmHg (lit.) |
| density 0.885 g/mL at 25 °C (lit.) | density 0.726 g/mL at 25 °C (lit.) | density 0.94 g/mL at 25 °C (lit.) | density 0.899 g/mL at 25 °C (lit.) |
| form liquid | form liquid | form liquid | form liquid |
| storage temp. 2-8°C | storage temp. 2-8°C | storage temp. 2-8°C | storage temp. 2-8°C |
General description
Application
accessory
signalword
Warning
hcodes
Hazard Classifications
Flam. Liq. 3
Storage Class
3 - Flammable liquids
wgk
WGK 3
flash_point_f
138.2 °F - closed cup
flash_point_c
59 °C - closed cup
ppe
Eyeshields, Gloves, multi-purpose combination respirator cartridge (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 212830-1G | 04061838772350 |
| 212830-5G | 04061838772367 |
Active Filters
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service