All Photos(1)
About This Item
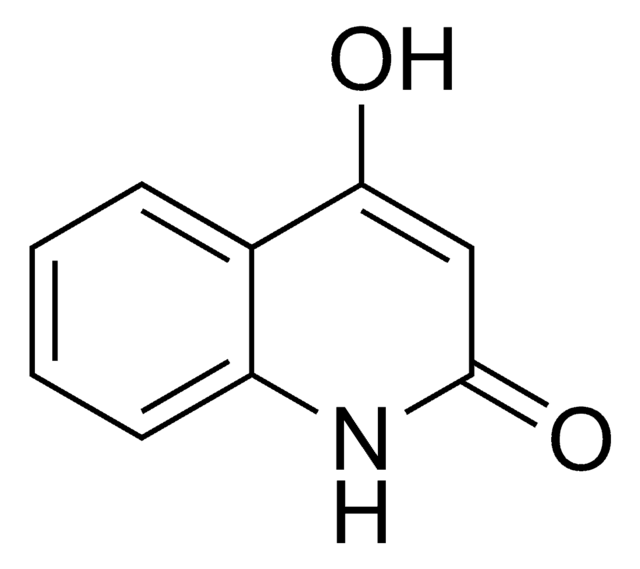
Empirical Formula (Hill Notation):
C10H9NO2
CAS Number:
Molecular Weight:
175.18
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
assay
98%
form
powder
mp
269-271 °C (lit.)
SMILES string
CN1C(=O)C=C(O)c2ccccc12
InChI
1S/C10H9NO2/c1-11-8-5-3-2-4-7(8)9(12)6-10(11)13/h2-6,12H,1H3
InChI key
RTNPPPQVXREFKX-UHFFFAOYSA-N
Application
4-Hydroxy-1-methyl-2(1H)-quinolone was used as nucleophile in electrochemical oxidation of catechols and the reaction was studied by cyclic voltammetry and controlled-potential coulometry. It was used in ceric ammonium nitrate-mediated oxidative cycloaddition of 1,3-dicarbonyls to conjugated compounds to yield substituted dihydrofurans, dihydrofurocoumarins, dihydrofuroquinolinones and dihydrofurophenalenones. It was used in the synthesis of :
- 3-(4-methoxybenzyl)-4-hydroxy-1-methylquinolinone
- 3-(benzo[d][1,3]dioxol-5-ylmethyl)-4-hydroxy-1-methylquinolin-2(1H)-one
- 3-(4-chlorobenzyl)-4-hydroxy-1-methylquinolin-2(1H)-one
- 3,3′-bis(4-chlorobenzylidene)-1,10-methylquinolin-2,20-(1H)-one
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Mechanistic study of electrochemical oxidation of catechols in the presence of 4-hydroxy-1-methyl-2 (1H)-quinolone: Application to the electrochemical synthesis.
Fakhari AR, et al.
Electrochimica Acta, 50(27), 5322-5328 (2005)
Iridium catalysed alkylation of 4-hydroxy coumarin, 4-hydroxy-2-quinolones and quinolin-4 (1H)-one with alcohols under solvent free thermal conditions.
Grigg R, et al.
Tetrahedron, 65(36), 7468-7473 (2009)
Ceric ammonium nitrate (CAN)-mediated oxidative cycloaddition of 1, 3-dicarbonyls to conjugated compounds. Efficient synthesis of dihydrofurans, dihydrofurocoumarins, dihydrofuroquinolinones, dihydrofurophenalenones, and furonaphthoquinone natural products.
Lee YR, et al.
Tetrahedron, 56(45), 8845-8853 (2000)
Michael B Hicks et al.
The Analyst, 142(3), 525-536 (2017-01-18)
The use of a coulometric array detector in tandem with HPLC-UV was evaluated for the absolute quantitation of pharmaceutical compounds without standards, an important capability gap in contemporary pharmaceutical research and development. The high-efficiency LC flow-through electrochemical detector system allows
Mohankumar Saraladevi Resmi et al.
FEBS letters, 589(3), 332-341 (2015-01-06)
Type III polyketide synthases (PKSs) catalyze the biosynthesis of various medicinally important secondary metabolites in plants, but their role in growth and stress response is unclear. Here, we overexpressed quinolone synthase (QNS) from bael in tobacco. QNS-overexpressing plants showed an
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service