903981
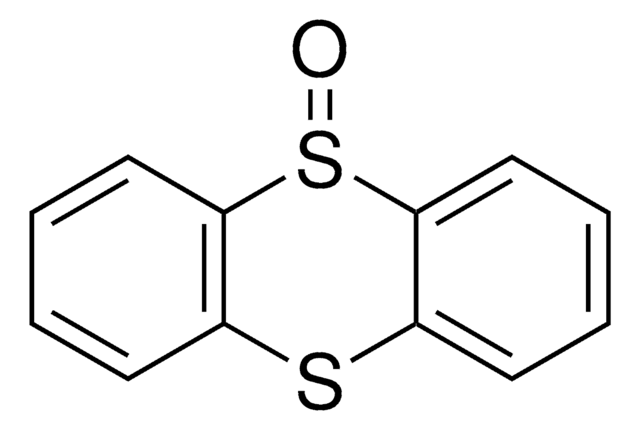
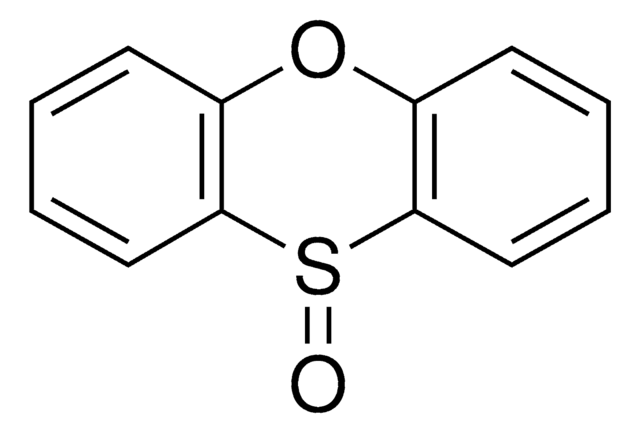
2,3,7,8-Tetrafluorothianthrene-S-oxide
≥95%
Synonym(s):
2,3,7,8-Tetrafluorothianthrene-5-oxide, Fluorinated sulfoxide-based thianthrene reagent, Ritter C-H functionalization linchpin, TFT S-oxide, TFT-reagent
About This Item
Recommended Products
Assay
≥95%
form
solid
mp
253 °C
functional group
fluoro
sulfoxide
thioether
storage temp.
−20°C
Related Categories
Application
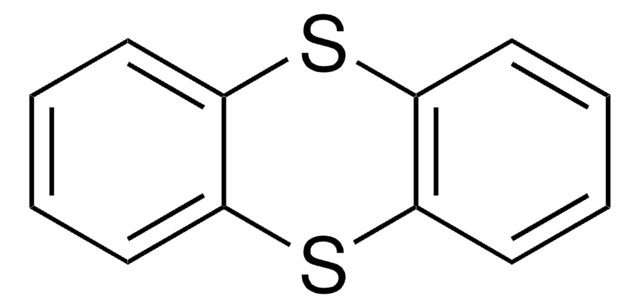
The nonfluorinated thianthrene S-oxide is also available as catalog# 903973 for all arenes more electron-rich than anisole.
Other Notes
Legal Information
This product is manufactured pursuant to a license with Studiengesellschaft Kohle mbH
related product
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Related Content
The Ritter lab currently focuses on fluorination chemistry for late-stage functionalization of complex natural and unnatural products. PhenoFluor™ has been developed as a general reagent for the selective, predictable, direct deoxyfluorination of complex alcohols and phenols.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service