All Photos(1)
About This Item
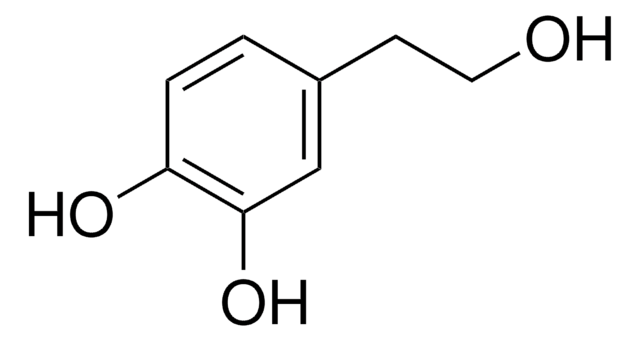
Linear Formula:
(HO)2C6H3CH(OH)CO2H
CAS Number:
Molecular Weight:
184.15
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
95%
mp
136-137 °C (dec.) (lit.)
functional group
carboxylic acid
hydroxyl
SMILES string
OC(C(O)=O)c1ccc(O)c(O)c1
InChI
1S/C8H8O5/c9-5-2-1-4(3-6(5)10)7(11)8(12)13/h1-3,7,9-11H,(H,12,13)
InChI key
RGHMISIYKIHAJW-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Metabolite of norepinephrine.
Application
DL-3,4-Dihydroxymandelic acid was used in the simultaneous analysis of 4-hydroxy-3-methoxymandelic acid and 4-hydroxy- 3-methoxyphenylacetic acid in urine. It was also used to study the changes in body temperature.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
M Kawamura et al.
Journal of the autonomic nervous system, 66(3), 145-148 (1997-12-24)
After norepinephrine (NE) is deaminated by monoamine oxidase (MAO), the aldehyde formed is either metabolized to 3,4-dihydroxy-mandelic acid (DHMA) by aldehyde dehydrogenase or is converted to 3,4-dihydroxyphenylglycol (DHPG) by aldehyde or aldose reductase. The present study examined the effects of
J Y Li et al.
Analytical biochemistry, 190(2), 354-359 (1990-11-01)
A quantitative assay for the diphenol oxidase activity of tyrosinase (EC 1.14.18.1) using high-pressure liquid chromatography with electrochemical detection is described. The assay is based on the observation (M. Sugumaran, 1986, Biochemistry 25, 4489-4492) that tyrosinase catalyzes the oxidative decarboxylation
S J Soldin et al.
Clinical chemistry, 26(2), 291-294 (1980-02-01)
We describe a rapid, reliable "high-performance" liquid-chromatographic method of simultaneously analyzing for 4-hydroxy-3-methoxymandelic acid (I) and 4-hydroxy-3-methoxyphenylacetic acid (II) in urine. Paired-ion chromatography and amperometric detection are used in the method. A 5-mL aliquot of urine is adjusted to pH
Andrea E Schwaninger et al.
Drug metabolism and disposition: the biological fate of chemicals, 39(11), 1998-2002 (2011-07-29)
Different pharmacokinetic properties are known for the two enantiomers of the entactogen 3,4-methylendioxy-methamphetamine (MDMA), most likely due to enantioselective metabolism. The aim of the present work was 1) the investigation of the main sulfotransferases (SULT) isoenzymes involved in the sulfation
T H Czapla et al.
Biochimica et biophysica acta, 1077(3), 400-406 (1991-04-29)
Cyclic voltammetric and chronoamperometric data are consistent with a process in which 3,4-dihydroxymandelic acid (DOMA) is oxidized initially in a two-electron step to its corresponding o-benzoquinone. This species is unstable and undergoes the rate-determining loss of CO2 (k = 1.6
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service