59770
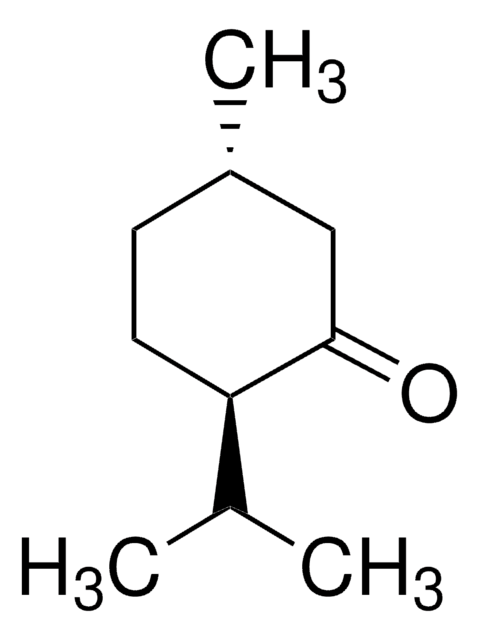
(−)-Isopulegol
analytical standard
Synonym(s):
(1R,2S,5R)-2-Isopropenyl-5-methylcyclohexanol, (1R,3R,4S)-p-Menth-8-en-3-ol
About This Item
Recommended Products
grade
analytical standard
Quality Level
Assay
≥99.0% (sum of enantiomers, GC)
optical activity
[α]20/D −22±0.5°, neat
optical purity
enantiomeric ratio: ≥99.5:0.5 (GC)
shelf life
limited shelf life, expiry date on the label
technique(s)
HPLC: suitable
gas chromatography (GC): suitable
refractive index
n20/D 1.471 (lit.)
n20/D 1.472
bp
212 °C (lit.)
density
0.912 g/mL at 25 °C (lit.)
application(s)
cleaning products
cosmetics
flavors and fragrances
food and beverages
personal care
format
neat
SMILES string
C[C@@H]1CC[C@H]([C@H](O)C1)C(C)=C
InChI
1S/C10H18O/c1-7(2)9-5-4-8(3)6-10(9)11/h8-11H,1,4-6H2,2-3H3/t8-,9+,10-/m1/s1
InChI key
ZYTMANIQRDEHIO-KXUCPTDWSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- Complex commercial sample matrices by gas chromatography coupled to vacuum ultraviolet detector (GC-VUV).
- Mentha piperita and M. arvensis essential oils by enantioselective GC.
- Urine samples by high performance liquid chromatography combined with tandem mass spectrometry (HPLC-MS/MS).
Other Notes
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
10 - Combustible liquids
WGK
WGK 1
Flash Point(F)
172.4 °F - closed cup
Flash Point(C)
78 °C - closed cup
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service