All Photos(2)
About This Item
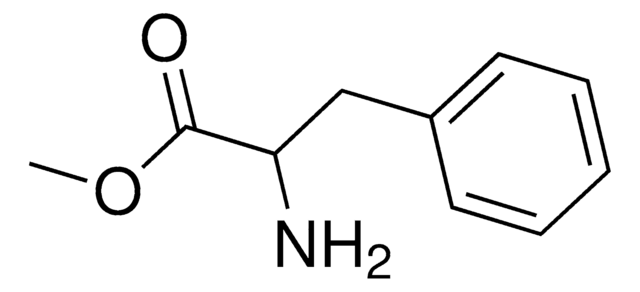
Linear Formula:
C6H5CH2CH(NH2)CO2CH3 · HCL
CAS Number:
Molecular Weight:
215.68
MDL number:
UNSPSC Code:
12352209
eCl@ss:
32160406
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
reaction suitability
reaction type: solution phase peptide synthesis
mp
159-163 °C (lit.)
application(s)
peptide synthesis
storage temp.
2-8°C
SMILES string
Cl[H].COC(=O)[C@H](N)Cc1ccccc1
InChI
1S/C10H13NO2.ClH/c1-13-10(12)9(11)7-8-5-3-2-4-6-8;/h2-6,9H,7,11H2,1H3;1H/t9-;/m1./s1
InChI key
SWVMLNPDTIFDDY-SBSPUUFOSA-N
Looking for similar products? Visit Product Comparison Guide
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Shotaro Tsuchiyama et al.
Biotechnology progress, 23(4), 820-823 (2007-05-08)
The PST-01 protease is a metalloprotease that has zinc ion at the active center and is very stable in the presence of water-soluble organic solvents. The reaction rates and the equilibrium yields of the aspartame precursor N-carbobenzoxy-L-aspartyl-L-phenylalanine methyl ester (Cbz-Asp-Phe-OMe)
Ikuo Kira et al.
Journal of bioscience and bioengineering, 108(3), 190-193 (2009-08-12)
Screening was carried out for microorganisms able to produce N-(l-alpha-l-aspartyl)-l-phenylalanine methyl ester [APM] from l-isoasparagine and l-phenylalanine methyl ester hydrochloride. Of the 422 strains examined, 44 strains belonging to the family Enterobacteriaceae were found to produce APM. The enzyme catalyzing
C S Rosenfeld
Blood, 80(9), 2401-2405 (1992-11-01)
Phenylalanine methylester (PME), a lysosomotropic compound can be used to deplete monocytes and myeloid cells from peripheral blood and bone marrow (BM). The potential of PME for purging leukemic cells from BM was investigated using U937 and HL-60 cell lines
R D Skwierczynski et al.
Pharmaceutical research, 10(8), 1174-1180 (1993-08-01)
The kinetics of demethylation of aspartame and L-phenylalanine methyl ester were studied in aqueous solution at 25 degrees C over the pH range 0.27-11.5. The pseudo-first-order rate constant for aspartame was resolved into individual contributions from methyl ester hydrolysis and
G D Castro et al.
Research communications in molecular pathology and pharmacology, 98(1), 85-90 (1998-01-22)
Reaction mixtures containing phenylalanine methyl ester and thymine in pure carbon tetrachloride in the presence of benzoyl peroxide produced trichloromethyl and trichloromethylperoxyl free radicals which via hydrogen abstraction reactions sparked the formation of phenylalanine-thymine adducts, whose structures were elucidated by
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service