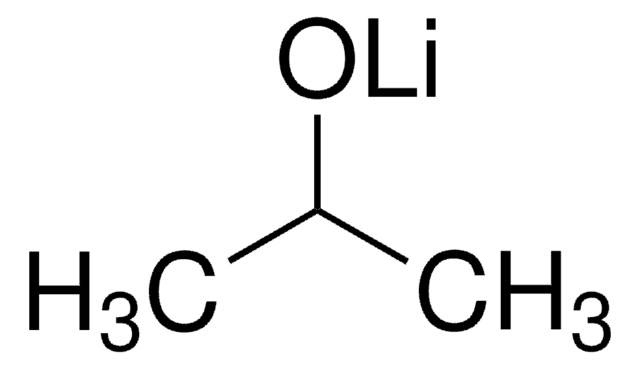
About This Item
Recommended Products
Assay
97%
form
powder and chunks
SMILES string
[Li+].CC(C)(C)[O-]
InChI
1S/C4H9O.Li/c1-4(2,3)5;/h1-3H3;/q-1;+1
InChI key
LZWQNOHZMQIFBX-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
Other synthetic applications:
- In combination with potassium diisopropylamide, LiOtBu can be used to deprotonate 1-(phenylseleno) alkenes and bis (phenylseleno) acetals.
- LiOtBu can mediate the α-alkylation reaction of ketones with primary alcohols in the absence of any transition metal catalyst.
- LiOtBu is an effective base for the synthesis of 3,4,5-trisubstituted 3H-oxazol-2-ones and 3,4-disubstituted (Z)-oxazolidin-2-ones from substituted propargyl alcohols and aryl/alkyl isocyanates using DMF as a solvent.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Self-heat. 1 - Skin Corr. 1B
Supplementary Hazards
Storage Class Code
4.2 - Pyrophoric and self-heating hazardous materials
WGK
WGK 3
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Few Monolayer Atomic Layer Deposition (ALD) on Surfaces and Interfaces for Energy Applications
Nanomaterials are considered a route to the innovations required for large-scale implementation of renewable energy technologies in society to make our life sustainable.
Nanomaterials are considered a route to the innovations required for large-scale implementation of renewable energy technologies in society to make our life sustainable.
Nanomaterials are considered a route to the innovations required for large-scale implementation of renewable energy technologies in society to make our life sustainable.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service