L2037
β-Lapachone
≥98% (TLC)
Synonyme(s) :
ARQ 501, NSC 26326, NSC 629749, SL 11001
Se connecterpour consulter vos tarifs contractuels et ceux de votre entreprise/organisme
About This Item
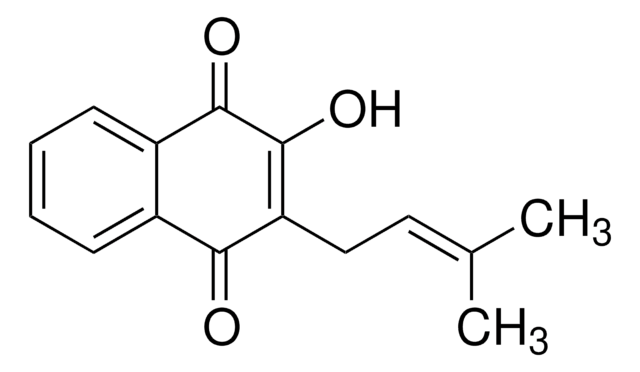
Formule empirique (notation de Hill) :
C15H14O3
Numéro CAS:
Poids moléculaire :
242.27
Numéro MDL:
Code UNSPSC :
12352200
ID de substance PubChem :
Nomenclature NACRES :
NA.77
Produits recommandés
Source biologique
synthetic (organic)
Essai
≥98% (TLC)
Forme
powder
Chaîne SMILES
CC1(C)CCC2=C(O1)c3ccccc3C(=O)C2=O
InChI
1S/C15H14O3/c1-15(2)8-7-11-13(17)12(16)9-5-3-4-6-10(9)14(11)18-15/h3-6H,7-8H2,1-2H3
Clé InChI
QZPQTZZNNJUOLS-UHFFFAOYSA-N
Application
β-Lapachone has been used:
- as an anticancer compound in catalase-inhibitable luminol/hydrogen peroxide (HRP)-dependent chemiluminometric assay in Lewis lung carcinoma (LLC) cells and isolated mitochondria
- as a naphthoquinone to study its effects on the growth and differentiation of mice granulocyte and macrophage progenitor cells
- as a substrate to study the enzyme activity of human recombinant NAD(P)H dehydrogenase 1 (NQO1) protein
Actions biochimiques/physiologiques
β-Lapachone acts as a DNA topoisomerase type I inhibitor. It exhibits anti-fungal, anti-bacterial, trypanocidal, and antiviral properties. β-Lapachone also inhibits nitric oxide (NO) and inducible NO synthase (iNOS) in alveolar macrophages.
β-Lapachone is a naturally occurring quinone obtained from the bark of the lapacho tree (Tabebuia avellanedae) with cancer chemopreventive properties. Induces apoptosis in HL-60 and human prostate cancer cells.
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Équipement de protection individuelle
Eyeshields, Gloves, type N95 (US)
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Cleverson Neves-Pinto et al.
Journal of medicinal chemistry, 45(10), 2112-2115 (2002-05-03)
An intensive effort has been directed toward finding alternative drugs for treatment of Chagas' disease, caused by Trypanosoma cruzi, and prophylaxis of blood in endemic areas. Our research comprises the synthesis and trypanocidal screening of derivatives from naphthoquinones. Herein a
Byung Tae Choi et al.
Anti-cancer drugs, 14(10), 845-850 (2003-11-05)
beta-Lapachone is a naturally occurring quinone obtained from the bark of the lapacho tree (Tabebuia avellanedae) with cancer chemopreventive properties. The objective of the present study was to investigate the effect of beta-lapachone on the cell growth and apoptosis in
Carla Ríos-Luci et al.
European journal of medicinal chemistry, 53, 264-274 (2012-05-09)
In this study, we describe the synthesis of a series of α- and β-lapachone containing hydroxyl or methoxyl groups on the benzene ring, by means of the selective acid promoted cyclization of the appropriate lapachol analog. The evaluation of the
Gang Huang et al.
Theranostics, 3(2), 116-126 (2013-02-21)
Superparamagnetic iron oxide nanoparticles (SPION) are an important and versatile nano- platform with broad biological applications. Despite extensive studies, the biological and pharmacological activities of SPION have not been exploited in therapeutic applications. Recently, β-lapachone (β-lap), a novel anticancer drug
Dong-Oh Moon et al.
International immunopharmacology, 7(4), 506-514 (2007-02-27)
beta-Lapachone (LAPA) is a chemotherapeutic agent that can inhibit the expression of nitric oxide (NO) and inducible NO synthase (iNOS) in alveolar macrophages. No other information on the agent's anti-inflammatory activity has been reported. In the present study, we investigated
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique