ROAPRO
Roche
Aprotinin
from bovine lung
Synonyme(s) :
Aprotinin, pancreatic trypsin inhibitor, trypsin inhibitor, pancreas type (bpti), trypsin-kallikrein inhibitor
About This Item
Produits recommandés
Source biologique
bovine lung
Niveau de qualité
Forme
lyophilized
Conditionnement
pkg of 10 mg (10236624001)
pkg of 100 mg (11583794001)
pkg of 50 mg (10981532001)
Fabricant/nom de marque
Roche
Technique(s)
electrophoresis: suitable
tissue culture: suitable
Plage de pH
3-10
Solubilité
water: soluble 10 mg/mL
Absorption
0.84 at 280 nm
Conditions d'expédition
wet ice
Température de stockage
2-8°C
Description générale
Spécificité
Cathepsin G, acrosin, human leukocyte elastase, and human urokinase are weakly inhibited. Factor Xa, thrombin, subtilisin, papain, pepsin, angiotensin-converting enzyme (ACE), carboxypeptidase A and B, other metalloproteases, and thiolproteases are not inhibited.
Application
- Further applications: Purification of urokinase, trypsin, and chymotrypsin on immobilized aprotinin
- Quantification of kallikrein activity in mixtures of esterases and proteases
- Controlled degradation of substrates by avoiding nonspecific proteolysis in clinical chemical tests
- Aprotinin as a model protein in protein-folding studies
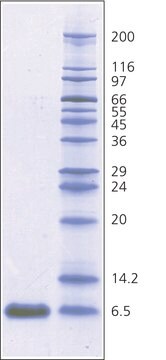
- Molecular weight marker in SDS-polyacrylamide gel electrophoresis
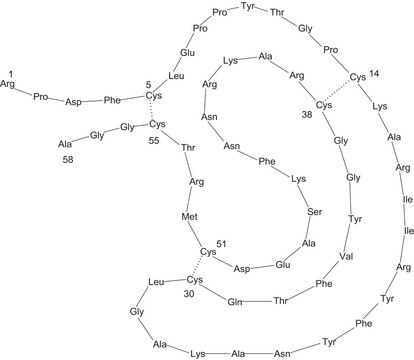
Séquence
Définition de l'unité
One inhibitor unit (IU) (+25 °C, BAEE as substrate) corresponds to about 2.8 inhibitor units (+25 °C, Chromozym TRY as substrate).
One inhibitor unit (IU) (+25 °C, BAEE as substrate) corresponds to about 26 kallikrein inhibitor units (KIU) (+25 °C).
One inhibitor unit (IU) (+25 °C, BAEE as substrate) corresponds to about 0.067 inhibitor units (+25 °C; Bz-D,L-Arg-4-Na as substrate, trypsin determination at pH 7.8).
One kallikrein inhibitor unit = 0.17 μg crystalline aprotinin.
Notes préparatoires
Working solution: Soluble in water (10 mg/ml) or aqueous buffer solution (e.g., 0.1 M Tris, pH 8.0).
Note: To avoid adsorption of aprotinin onto negatively charged solid phases, e.g., chromatography gels, ultrafiltration membranes, the NaCl concentration should be above 0.1 M or other suitable salts should be added to all buffers used during the separation.
Storage conditions (working solution): -15 to -25 °C
Reconstitution
Aliquots stored at -15 to -25 °C are stable for approximately 6 months.
Note: Avoid repeated freezing and thawing and exposure to strongly alkaline solutions (inactive at pH > 12.8).
Autres remarques
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 1
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Kinase and Phosphatidylinositide 3-Kinase/Akt Signaling
fractional diagonal chromatography
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique