409006
Di(ethylene glycol) dimethacrylate
95%, cross-linking reagent polymerization reactions, methacrylate, 300 ppm monomethyl ether hydroquinone as inhibitor
Synonym(s):
2,2′-Oxybisethanol dimethacrylate, 2,2′-Oxydiethyl dimethacrylate, Diethylene glycol, dimethacrylate, Polyethylene glycol
About This Item
Recommended Products
product name
Di(ethylene glycol) dimethacrylate, 95%
Quality Level
Assay
95%
form
liquid
contains
300 ppm monomethyl ether hydroquinone as inhibitor
reaction suitability
reagent type: cross-linking reagent
reaction type: Polymerization Reactions
refractive index
n20/D 1.458 (lit.)
bp
134 °C/2 mmHg (lit.)
density
1.082 g/mL at 25 °C (lit.)
Ω-end
methacrylate
α-end
methacrylate
polymer architecture
shape: linear
functionality: homobifunctional
SMILES string
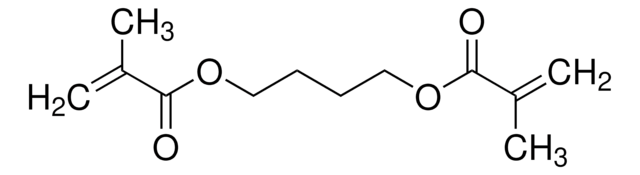
CC(=C)C(=O)OCCOCCOC(=O)C(C)=C
InChI
1S/C12H18O5/c1-9(2)11(13)16-7-5-15-6-8-17-12(14)10(3)4/h1,3,5-8H2,2,4H3
InChI key
XFCMNSHQOZQILR-UHFFFAOYSA-N
Application
- Urethane dimethacrylate-based photopolymerizable resins for stereolithography 3D printing: A physicochemical characterisation and biocompatibility evaluation.: This study explores the use of urethane dimethacrylate-based resins in stereolithography 3D printing. It includes an in-depth physicochemical characterization and assesses the biocompatibility of the materials, highlighting their potential for medical and dental applications (Pitzanti G et al., 2024).
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
10 - Combustible liquids
WGK
WGK 1
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service